ABSTRACT
Soil contamination by petroleum hydrocarbons and heavy metals threatens agricultural productivity and public health in semi-arid regions like Nigeria's Sudan Savannah. Phytoremediation using Jatropha curcas offers a sustainable strategy, yet the influence of propagation method on remediation efficiency remains underexplored. A 12-week pot experiment was conducted at Aliko Dangote University of Science and Technology, Wudil, using a Completely Randomized Design with three replications. The study evaluated phytoremediation potential of Jatropha curcas propagated by seed (JS) and stem cuttings (JC) in soils contaminated with two petroleum levels (1000 mL and 1500 mL per 12 kg soil). Unplanted contaminated controls were included. Total Petroleum Hydrocarbons (TPH) were analyzed using Gas Chromatography-Flame Ionization Detector, heavy metals (Pb, Cd, Zn, Cu) using Atomic Absorption Spectrophotometry, and soil physicochemical properties were assessed following standard protocols. Data were analyzed using ANOVA with means separated by Tukey's HSD test (p < 0.05). Jatropha curcas significantly reduced TPH compared to unplanted controls. Seed-propagated Jatropha achieved the lowest residual TPH (24,530.5 mg/kg at 1000 mL; 36,683.9 mg/kg at 1500 mL) and highest remediation efficiencies (62.4% and 62.7%), outperforming cutting-propagated plants (56.4% and 59.3%). Soil physicochemical properties improved significantly under vegetation, with JS-treated soils showing highest pH (7.11), organic carbon (1.88%), cation exchange capacity (5.80 cmol/kg), and available phosphorus (18.8 mg/kg), alongside reduced bulk density (1.59 g/cm³) compared to contaminated controls. Jatropha curcas demonstrates strong phytoremediation potential for petroleum-contaminated soils in the Sudan Savannah, with seed propagation proving superior to stem cuttings due to enhanced root development. The plant's ability to simultaneously restore soil fertility supports its dual role in environmental cleanup and land rehabilitation.
Keywords: Phytoremediation; Jatropha curcas; Petroleum Hydrocarbons; Heavy Metals; Sudan Savannah; Propagation Methods; Soil Restoration
INTRODUCTION
Environmental pollution resulting from industrialization, urbanization, and petroleum-based economic activities has led to significant contamination of soils by petroleum hydrocarbons and heavy metals across the globe. These contaminants threaten terrestrial ecosystems, reduce soil fertility, and jeopardize food safety and public health. According to the United Nations Environment Programme, over 35% of global soils are moderately to severely degraded due to anthropogenic activities, with petroleum spills and heavy metal accumulation among the leading causes [1]. Contaminated soils reduce agricultural productivity, alter microbial diversity, and cause long-term ecological harm [2-3].
Petroleum hydrocarbons are composed of complex organic compounds, including alkanes, aromatics, and polycyclic aromatic hydrocarbons (PAHs). These substances are hydrophobic, persistent, and toxic, often causing mutagenic and carcinogenic effects in plants and soil organisms. They are released into the environment through oil spills, pipeline leaks, vehicle servicing, and industrial effluents [4]. Furthermore, they affect soil structure, reduce nutrient availability, and impair microbial functions necessary for healthy soil [5-6]. Simultaneously, heavy metals such as cadmium (Cd), lead (Pb), chromium (Cr), and zinc (Zn) persist in soils without degradation. They originate from mining, agrochemicals, fossil fuel combustion, and waste dumping and accumulate in the soil-food chain, causing severe health effects such as carcinogenicity, organ failure, and neurotoxicity [7-8].
In Nigeria, contamination of soils by petroleum hydrocarbons and heavy metals has reached alarming levels, especially in urban and semi-urban regions where illegal refining, waste oil disposal, and use of contaminated water for irrigation are common [9-10]. Although the Niger Delta is well-known for oil pollution, northern regions like Wudil in Kano State, situated in the Sudan Savannah ecological zone, are increasingly affected by unregulated auto-mechanic workshops, fuel stations, and agricultural runoff [11-12]. The Sudan Savannah, characterized by sandy loam soils, sparse vegetation, and short rainy seasons, facilitates rapid contaminant infiltration and mobility [13]. These environmental conditions increase the vulnerability of local soils to both petroleum and heavy metal pollution.
Traditional methods for remediating polluted soils such as excavation, soil washing, vitrification, and chemical oxidation are often expensive, energy-intensive, and disruptive to the natural environment [14]. These limitations have necessitated the adoption of phytoremediation, a cost-effective, green, and sustainable technique that uses selected plants to stabilize, degrade, or extract contaminants from soil and water [15-16]. Phytoremediation leverages natural plant-microbe interactions and enzymatic activities in the rhizosphere to detoxify both organic and inorganic pollutants [17-18]. It is widely recognized as a promising solution for low- to medium-level contaminated sites, particularly in developing countries [3,19].
Among the various plant species studied for phytoremediation, Jatropha curcas L., a member of the Euphorbiaceae family, has attracted global attention. Native to Central America but now cultivated across Africa and Asia, Jatropha is drought-resistant, fast-growing, and capable of thriving on marginal lands with low fertility [20-21]. Studies have shown that Jatropha curcas possesses strong phytoremediation potential due to its ability to tolerate and accumulate both petroleum hydrocarbons and heavy metals, including Pb, Zn, and Cd [22-23]. Its extensive root system, high biomass yield, and phytochemical composition enhance its effectiveness in phytoextraction, phytostabilization, and phytodegradation processes [24-25]. Despite the proven ability of Jatropha curcas to survive in contaminated soils, there is limited comparative data on the phytoremediation efficiency of the plant when propagated by seed versus stem cuttings two commonly used methods in tropical agriculture. The method of propagation can influence root development, biomass production, pollutant uptake, and overall survival in polluted environments [26]. Without this knowledge, recommendations for scaling phytoremediation strategies using Jatropha curcas remain incomplete and potentially ineffective in practical settings. Furthermore, while several studies have been conducted on the potentials of Jatropha curcas for remediation of heavy metals and hydrocarbons in contaminated soils, none have compared seed versus cutting propagation methods under varying contamination levels in the Sudan Savannah zone of Nigeria. This study addresses this critical knowledge gap [27-30].
MATERIALS AND METHODS
Experimental Site
The research was conducted at the nursery site of the Department of Forestry, Aliko Dangote University of Science and Technology, Wudil, located in Wudil Local Government Area of Kano State, Nigeria (11°48′N, 8°50′E; elevation ~457 m above sea level). The area lies within the Sudan Savannah ecological zone, characterized by distinct dry and wet seasons. Mean annual rainfall ranges between 800–1000 mm (May–September), mean annual temperature is approximately 27°C, and relative humidity averages 30–40% in the dry season and up to 80% during the wet season (NIMET, 2023). Soils are predominantly alfisols and entisols, sandy loam to loamy in texture, with low organic matter, nitrogen, and cation exchange capacity [31-32].
Figure 1: Map of the study area showing ADUSTECH Wudil experimental site
Experimental Materials
Soil Collection and Preparation
Disturbed topsoil samples were collected from the 0–20 cm depth within the university premises using a soil auger. A total of 20 auger samples were taken randomly across the collection area and combined to form a composite sample. The composite soil was air-dried for seven days, crushed gently, and passed through a 2 mm mesh sieve to ensure uniformity and remove debris, stones, and plant residues. The sieved soil was thoroughly homogenized and divided into 17 experimental units, each receiving 12 kg of soil. The soil type was predominantly sandy loam, characteristic of the Sudan Savannah zone.
Petroleum Hydrocarbon
Fresh petroleum (premium motor spirit) was sourced from Gano Investment petrol station, Wudil. It was applied at two contamination levels: 1000 mL and 1500 mL per 12 kg of soil. Based on an average crude oil density of 0.85 g/mL, these volumes correspond to approximately 70,833 mg/kg and 106,250 mg/kg, respectively, simulating realistic oil pollution scenarios [33-34].
Planting Materials
Seeds of Jatropha curcas were sourced from healthy, disease-free mother plants in Gaya Local Government Area, Kano State. Stem cuttings consisting of semi-hardwood sections with an average height of 22.27 cm and girth of approximately 4.0 cm were harvested from healthy, mature Jatropha curcas plants, as recommended for optimal root initiation [35-36].
Experimental Containers
Standard plastic buckets weighing 0.40 kg with dimensions of 32 cm height, 106 cm upper diameter, and 69 cm lower diameter (7 L capacity) were perforated at the base to allow for drainage. Each bucket was filled with 12 kg of prepared soil.
Experimental Design and Treatments
The experiment was laid out in a 3 × 3 factorial arrangement using a Completely Randomized Design (CRD). Factor A was propagation method (unplanted control [J0], Jatropha from cutting [JC], Jatropha from seed [JS]). Factor B was petroleum contamination level (0 mL [P0], 1000 mL [P1000], 1500 mL [P1500]). The nine treatment combinations are described in Table 1.
|
Treatment Code |
Propagation Method |
Petroleum Dose |
Description |
|
A (J0×P0) |
Unplanted Control |
0 mL |
Uncontaminated soil, no Jatropha |
|
B (JS×P0) |
Seed |
0 mL |
Uncontaminated soil + Jatropha seed |
|
C (JC×P0) |
Cutting |
0 mL |
Uncontaminated soil + Jatropha cutting |
|
D (J0×P1000) |
Unplanted Control |
1000 mL |
Contaminated soil (1000 mL), no Jatropha |
|
E (J0×P1500) |
Unplanted Control |
1500 mL |
Contaminated soil (1500 mL), no Jatropha |
|
F (JS×P1000) |
Seed |
1000 mL |
Contaminated soil (1000 mL) + Jatropha seed |
|
G (JS×P1500) |
Seed |
1500 mL |
Contaminated soil (1500 mL) + Jatropha seed |
|
H (JC×P1000) |
Cutting |
1000 mL |
Contaminated soil (1000 mL) + Jatropha cutting |
|
I (JC×P1500) |
Cutting |
1500 mL |
Contaminated soil (1500 mL) + Jatropha cutting |
Table 1: Treatment Combinations
Treatments F–I were replicated three times, while controls (A–E) were not replicated, resulting in a total of 17 experimental units. Measured volumes of petroleum were thoroughly mixed with the soil in each bucket and allowed to equilibrate for 7 days prior to planting to ensure uniform hydrocarbon dispersion [37]. After equilibration, seeds and cuttings were planted directly into the treated soil. The experiment was maintained for 12 weeks with regular irrigation using borehole water to maintain near-field capacity moisture.
Laboratory Analyses
Soil Sample Collection and Preparation
At the end of the 12-week experimental period, soil samples were collected from each experimental unit at the root zone. Samples were air-dried, gently crushed, sieved through a 2 mm mesh for physical and chemical analysis, and through a 0.5 mm mesh for TPH and heavy metal analyses. Samples were stored in labeled, airtight polythene bags at 4°C until analysis [38-39].
Total Petroleum Hydrocarbon (TPH) Determination
TPH concentration was determined using Gas Chromatography-Flame Ionization Detector (GC-FID) following extraction with n-hexane, according to USEPA Method 8015B [39]. Results were expressed in mg/kg dry soil. Remediation efficiency (%) was calculated as:
Remediation Efficiency (%) = C0−Ct/C0×100
Where C0 = initial TPH concentration and Ct = TPH concentration after phytoremediation.
Heavy Metal Analysis
Concentrations of lead (Pb), cadmium (Cd), zinc (Zn), and copper (Cu) were analyzed using Atomic Absorption Spectrophotometry (AAS) after wet digestion with a mixture of nitric acid (HNO₃) and perchloric acid (HClO₄), following AOAC methods [40]. Results were expressed in mg/kg dry weight.
Soil Physicochemical Properties
The following soil physicochemical properties were assessed using standard methods: soil pH potentiometrically in a 1:2.5 soil-to-deionized water suspension; electrical conductivity (EC) using a conductivity meter in a 1:2.5 soil-to-water extract [41]; organic carbon (OC) by Walkley-Black wet oxidation [43]; total nitrogen (N) by Macro-Kjeldahl digestion method [43]; available phosphorus (P) by Bray-1 method [44]; cation exchange capacity (CEC) by ammonium acetate (1N NH₄OAc) method at pH 7.0 [45]; bulk density (BD) by core method [43]; porosity (PR) calculated as [1−(BD/PD)]×100 assuming particle density (PD) of 2.65 g/cm³; and mean weight diameter (MWD) by wet-sieving method [40].
Statistical Analysis
Data were subjected to two-way Analysis of Variance (ANOVA) for a Completely Randomized Design using SAS software. Where significant differences were observed (p < 0.05), means were separated using Tukey's Studentized Range (HSD) test at 5% probability [46]. Results are presented as mean ± standard error.
RESULTS
Total Petroleum Hydrocarbon (TPH) Concentration and Remediation Efficiency
The effects of Jatropha curcas propagation method and petroleum contamination level on TPH concentration and remediation efficiency are presented in Table 2. ANOVA revealed that propagation method significantly (p < 0.05) influenced final TPH concentration, while contamination level and its interaction with propagation method were not significant. The unplanted control (J0) recorded the highest TPH concentrations: 65,217.1 ± 6483.43 mg/kg at P1000 and 98,490.4 ± 10,030.39 mg/kg at P1500, compared to 23.1 ± 14,460.14 mg/kg in the uncontaminated control (P0). This indicates negligible natural degradation in the absence of vegetation. Cutting-propagated Jatropha (JC) reduced TPH concentrations to 28,430.4 ± 421.98 mg/kg (P1000) and 40,104.0 ± 485.75 mg/kg (P1500), representing remediation efficiencies of 56.4% and 59.3%, respectively. Seed-propagated Jatropha (JS) achieved the greatest reductions: 24,530.5 ± 356.14 mg/kg (P1000) and 36,683.9 ± 367.57 mg/kg (P1500), corresponding to efficiencies of 62.4% and 62.7%. These values were significantly different (p < 0.05) from both the unplanted control and the JC treatments. The general trend indicates that TPH concentration increased with contamination level but decreased markedly under vegetation, with seed-propagated plants consistently outperforming cutting-propagated plants.
|
Trt. |
Pet. Dose (mg/kg) |
Mean TPH Conc. (mg/kg) |
Rem. Eff. (%) |
Phyto. Pot. |
|
J0 |
0 (P0) |
23.1 ± 14460.14 g |
(Control) |
Control |
|
85,000 mg/kg (P1000) |
65217.1 ± 6483.43 b |
- |
Low |
|
|
127,500 mg/kg (P1500) |
98490.4 ± 10030.39 a |
- |
Low |
|
|
JC |
0 (P0) |
21.7 ± 5953.99 g |
(Control) |
Control |
|
85,000 mg/kg (P1000) |
28430.4 ± 421.98 e |
56.4 |
Moderate |
|
|
127,500 mg/kg (P1500) |
40104.0 ± 485.75 c |
59.3 |
Moderate |
|
|
JS |
0 (P0) |
20.1 ± 5393.24 g |
(Control) |
Control |
|
85,000 mg/kg (P1000) |
24530.5 ± 356.14 f |
62.4 |
High |
|
|
127,500 mg/kg (P1500) |
36683.9 ± 367.57 d |
62.7 |
High |
Table 2: Mean TPH Concentration and Phytoremediation Efficiency of Jatropha curcas Under Different Treatments
Note: Trt. = Treatment; Pet. = Petroleum; TPH = Total Petroleum Hydrocarbon; Rem. Eff. = Remediation Efficiency; Phyto. Pot. = Phytoremediation Potential. J0 = Unplanted Control; JC = Jatropha from Cutting; JS = Jatropha from Seed
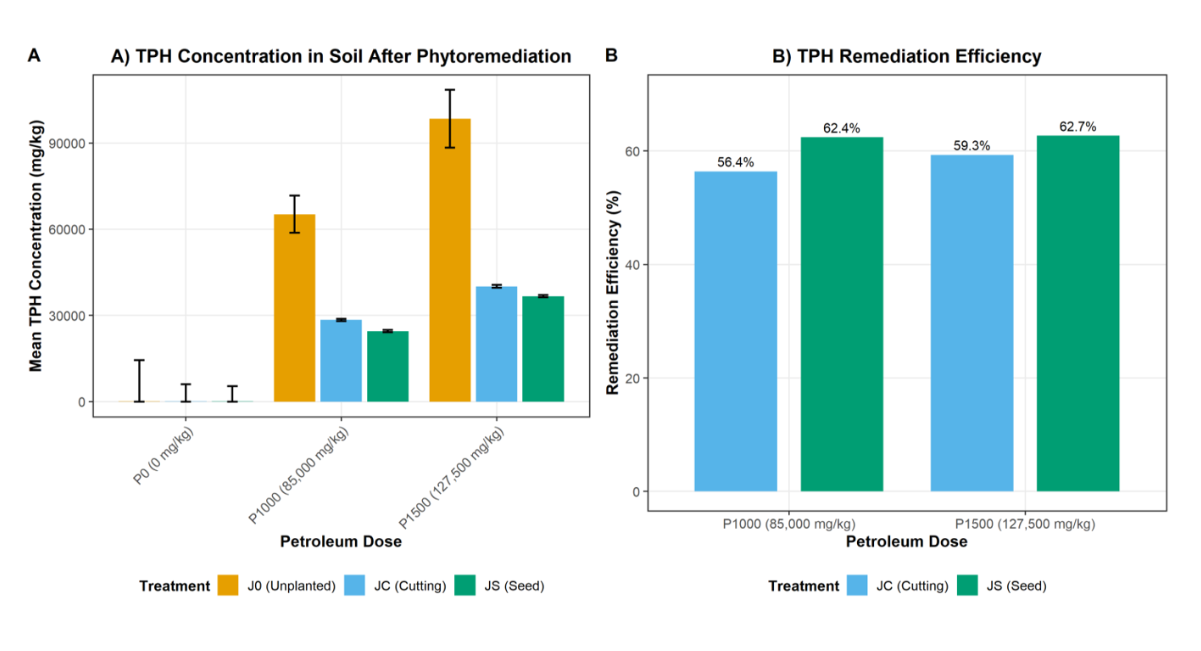
Figure 1: TPH Remediation Efficiency of Different Jatropha curcas Propagation Methods at Two Contamination Levels
Caption: Figure 1: Mean TPH concentrations (mg/kg) in soils after 12 weeks of phytoremediation under different treatments. J0 = Unplanted Control, JC = Jatropha from Cutting, JS = Jatropha from Seed. Error bars represent standard error of the mean. Bars with different letters are significantly different at p < 0.05 (Tukey's HSD).
Heavy Metal Concentrations
Post-remediation heavy metal concentrations varied significantly (p < 0.05) among treatments as shown in Table 3. Lead (Pb) concentrations in remediated soils ranged from 96.90 ± 1.04 mg/kg (JS×P1000) to 110.70 ± 0.72 mg/kg (JC×P1500), significantly lower than the unplanted controls (124.63–134.33 mg/kg). Cadmium (Cd) concentrations ranged from 3.20 ± 0.043 mg/kg (JS×P1000) to 3.79 ± 0.030 mg/kg (JC×P1500), representing significant reductions compared to unplanted controls (4.94–5.54 mg/kg). Zinc (Zn) concentrations ranged from 120.50 ± 1.25 mg/kg (JS×P1000) to 138.83 ± 0.78 mg/kg (JC×P1500), all significantly lower than unplanted controls (151.47–164.10 mg/kg). Copper (Cu) concentrations ranged from 83.93 ± 0.72 mg/kg (JS×P1000) to 94.20 ± 0.69 mg/kg (JC×P1500), compared to 104.37–114.83 mg/kg in unplanted controls. For all metals, seed-propagated treatments (JS) achieved lower residual concentrations than cutting-propagated treatments (JC), and both vegetated treatments were significantly lower than unplanted controls.
|
Treatment |
Zn (mg/kg) |
Pb (mg/kg) |
Cd (mg/kg) |
Cu (mg/kg) |
|
J0×P0 |
69.47 ± 0.58 g |
46.10 ± 0.55 g |
1.14 ± 0.020 e |
31.47 ± 0.41 g |
|
J0×P1000 |
151.47 ± 0.78 b |
124.63 ± 0.49 b |
4.94 ± 0.020 b |
104.37 ± 0.49 b |
|
J0×P1500 |
164.10 ± 0.75 a |
134.33 ± 0.40 a |
5.54 ± 0.020 a |
114.83 ± 0.55 a |
|
JC×P0 |
67.93 ± 0.66 g |
44.77 ± 0.61 g |
1.13 ± 0.023 e |
30.50 ± 0.49 g |
|
JC×P1000 |
126.70 ± 0.98 e |
101.13 ± 0.78 e |
3.38 ± 0.035 d |
87.10 ± 0.69 e |
|
JC×P1500 |
138.83 ± 0.78 c |
110.70 ± 0.72 c |
3.79 ± 0.030 c |
94.20 ± 0.69 c |
|
JS×P0 |
66.50 ± 0.76 g |
43.30 ± 0.69 g |
1.12 ± 0.020 e |
29.30 ± 0.46 g |
|
JS×P1000 |
120.50 ± 1.25 f |
96.90 ± 1.04 f |
3.20 ± 0.043 d |
83.93 ± 0.72 f |
|
JS×P1500 |
133.90 ± 1.01 d |
106.80 ± 0.92 d |
3.63 ± 0.038 c |
90.80 ± 0.75 d |
Table 3: Heavy Metal Concentrations (mg/kg) in Soil after Phytoremediation
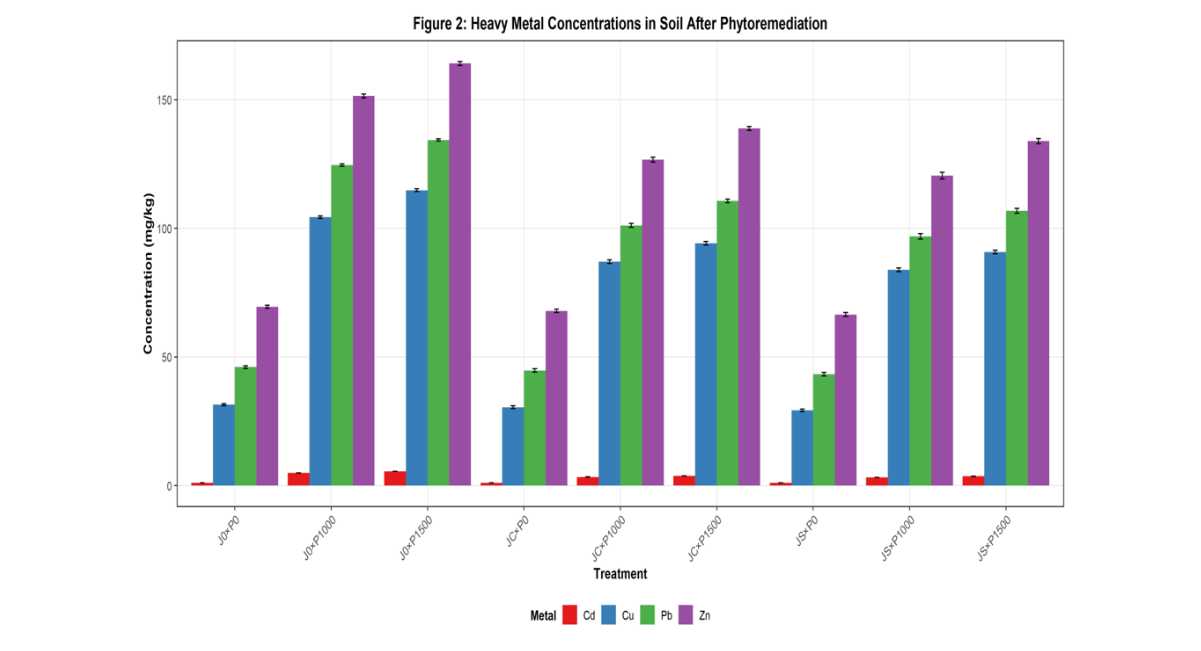
Figure 2: Heavy Metal Concentrations in Soils after Phytoremediation
Caption: Figure 2. Post-remediation concentrations of lead (Pb), cadmium (Cd), zinc (Zn), and copper (Cu) in soils under different treatments. J0 = Unplanted Control, JC = Jatropha from Cutting, JS = Jatropha from Seed. Values are means ± standard error. Different letters indicate significant differences at p < 0.05 (Tukey's HSD).
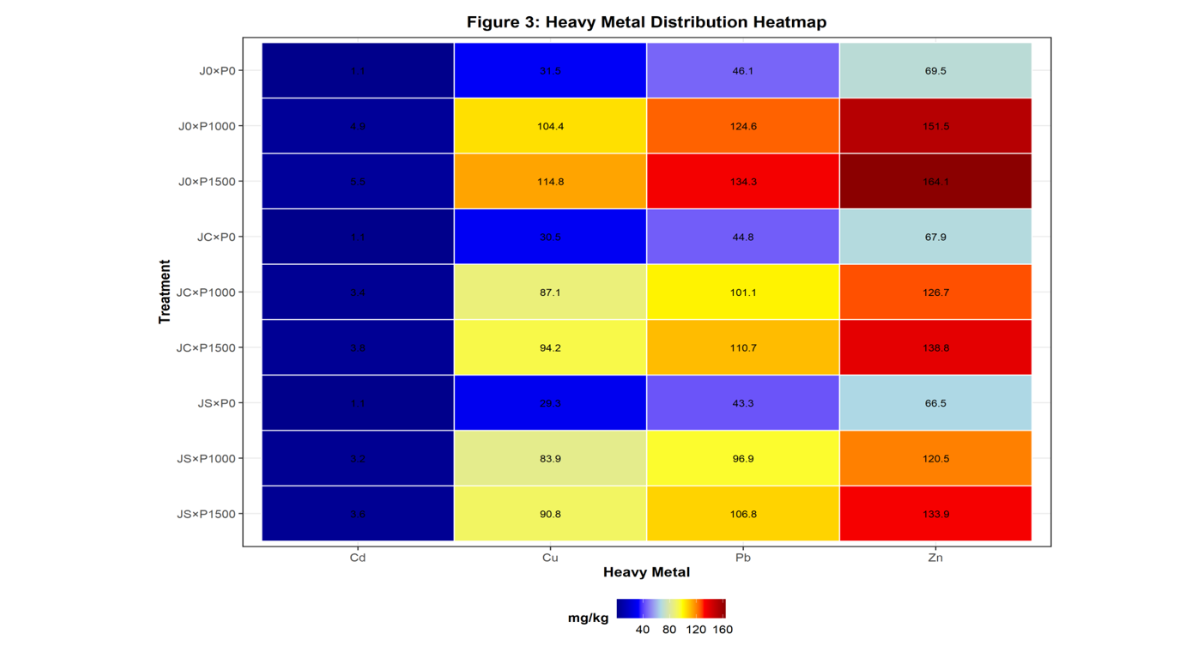
Figure 3: Heatmap of Heavy Metal Distribution across Treatments
Caption: Figure 3. Heatmap showing relative concentrations of heavy metals (Pb, Cd, Zn, Cu) across all treatment combinations. Darker colors indicate higher concentrations. Values are expressed in mg/kg
Soil Physicochemical Properties
The influence of Jatropha curcas propagation method and petroleum contamination on soil physicochemical properties is presented in Table 4. Petroleum contamination significantly reduced soil pH from 7.02 (J0×P0) to 6.25 (J0×P1500). Vegetation improved pH, with JS×P1500 reaching 6.65 and JC×P1500 reaching 6.58, compared to 6.25 in the unplanted control. The highest pH among contaminated vegetated treatments was recorded in JS×P1000 (6.82). Electrical conductivity (EC) increased with contamination level in unplanted controls (0.160 dS/m at P0 to 1.250 dS/m at P1500), but vegetation reduced EC, with JS×P1000 recording the lowest EC (0.477 dS/m) among contaminated treatments. Organic carbon (OC) decreased sharply with contamination in unplanted controls (2.39% at P0 to 1.06% at P1500). Vegetated treatments showed higher OC, with JS×P1000 (1.88%) and JS×P1500 (1.66%) outperforming JC treatments (1.73% and 1.53%, respectively). Total nitrogen (N) followed a similar trend, with JS×P1000 (0.200%) and JS×P1500 (0.162%) showing higher values than JC treatments (0.182% and 0.162%) and unplanted controls (0.120% and 0.095%). Available phosphorus (P) was significantly higher in vegetated treatments, with JS×P1000 (18.8 mg/kg) and JS×P1500 (16.3 mg/kg) compared to JC treatments (17.6 and 15.5 mg/kg) and unplanted controls (14.2 and 12.5 mg/kg). Cation exchange capacity (CEC) improved under vegetation, with JS×P1000 (5.80 cmol/kg) and JS×P1500 (5.10 cmol/kg) exceeding JC treatments (5.40 and 4.80 cmol/kg) and unplanted controls (4.20 and 3.80 cmol/kg). Bulk density (BD) increased with contamination in unplanted controls (1.413 to 1.823 g/cm³), but vegetated treatments showed lower BD, with JS×P1000 (1.577 g/cm³) and JS×P1500 (1.650 g/cm³) outperforming JC treatments (1.620 and 1.700 g/cm³). Porosity followed the inverse pattern. Mean weight diameter (MWD), an indicator of soil structural stability, decreased with contamination (1.003 mm at P0 to 0.480 mm at P1500 in unplanted controls). Vegetated treatments improved MWD, with JS×P1000 (0.753 mm) and JS×P1500 (0.650 mm) exceeding JC treatments (0.700 and 0.600 mm).
|
Treatment |
pH |
EC (dS/m) |
OC (%) |
N (%) |
P (mg/kg) |
K (cmol/kg) |
BD (g/cm³) |
Porosity (%) |
MWD (mm) |
CEC (cmol/kg) |
|
J0 × P0 |
7.02 ± 0.020 a |
0.160 ± 0.0058 g |
2.39 ± 0.023 a |
0.268 ± 0.0044 a |
25.13 ± 0.29 a |
0.274 ± 0.0035 a |
1.413 ± 0.0145 e |
47.43 ± 0.35 a |
1.003 ± 0.0203 a |
7.60 ± 0.12 a |
|
J0 × P1000 |
6.45 ± 0.017 d |
0.947 ± 0.0145 b |
1.26 ± 0.023 e |
0.120 ± 0.0029 e |
14.23 ± 0.20 c |
0.150 ± 0.0029 f |
1.750 ± 0.0173 b |
35.30 ± 0.96 e |
0.550 ± 0.0115 e |
4.20 ± 0.06 f |
|
J0 × P1500 |
6.25 ± 0.017 e |
1.250 ± 0.0173 a |
1.06 ± 0.023 f |
0.095 ± 0.0029 f |
12.53 ± 0.26 d |
0.125 ± 0.0029 f |
1.823 ± 0.0145 a |
32.53 ± 0.92 f |
0.480 ± 0.0115 f |
3.80 ± 0.06 f |
|
JC × P0 |
7.03 ± 0.018 a |
0.155 ± 0.0058 g |
2.42 ± 0.023 a |
0.273 ± 0.0044 a |
25.43 ± 0.29 a |
0.277 ± 0.0018 a |
1.393 ± 0.0145 e |
47.97 ± 0.43 a |
1.027 ± 0.0203 a |
7.70 ± 0.12 a |
|
JC × P1000 |
6.75 ± 0.017 b |
0.547 ± 0.0145 e |
1.73 ± 0.029 c |
0.182 ± 0.0044 c |
17.60 ± 0.26 b |
0.197 ± 0.0029 c |
1.620 ± 0.0173 d |
39.87 ± 0.52 c |
0.700 ± 0.0115 c |
5.40 ± 0.06 c |
|
JC × P1500 |
6.58 ± 0.017 c |
0.750 ± 0.0145 c |
1.53 ± 0.029 d |
0.162 ± 0.0029 d |
15.53 ± 0.26 c |
0.165 ± 0.0012 e |
1.700 ± 0.0115 c |
37.10 ± 0.38 d |
0.600 ± 0.0115 d |
4.80 ± 0.06 e |
|
JS × P0 |
7.05 ± 0.017 a |
0.150 ± 0.0058 g |
2.44 ± 0.023 a |
0.278 ± 0.0044 a |
25.70 ± 0.26 a |
0.280 ± 0.0018 a |
1.377 ± 0.0145 e |
48.47 ± 0.38 a |
1.050 ± 0.0173 a |
7.80 ± 0.12 a |
|
JS × P1000 |
6.82 ± 0.015 b |
0.477 ± 0.0145 f |
1.88 ± 0.020 b |
0.200 ± 0.0058 b |
18.80 ± 0.17 b |
0.215 ± 0.0015 b |
1.577 ± 0.0145 d |
41.50 ± 0.51 b |
0.753 ± 0.0145 b |
5.80 ± 0.06 b |
|
JS × P1500 |
6.65 ± 0.017 c |
0.677 ± 0.0145 d |
1.66 ± 0.023 c |
0.162 ± 0.0044 d |
16.30 ± 0.26 c |
0.180 ± 0.0012 d |
1.650 ± 0.0173 c |
38.50 ± 0.40 d |
0.650 ± 0.0115 c |
5.10 ± 0.06 d |
Table 4: Selected Soil Physicochemical Properties after the Experiment
Note: Means within a column followed by the same letter are not significantly different at p < 0.05 according to Tukey’s HSD test. EC = Electrical Conductivity; OC = Organic Carbon; N = Nitrogen; P = Phosphorus; K = Potassium; BD = Bulk Density; MWD = Mean Weight Diameter; CEC = Cation Exchange Capacity
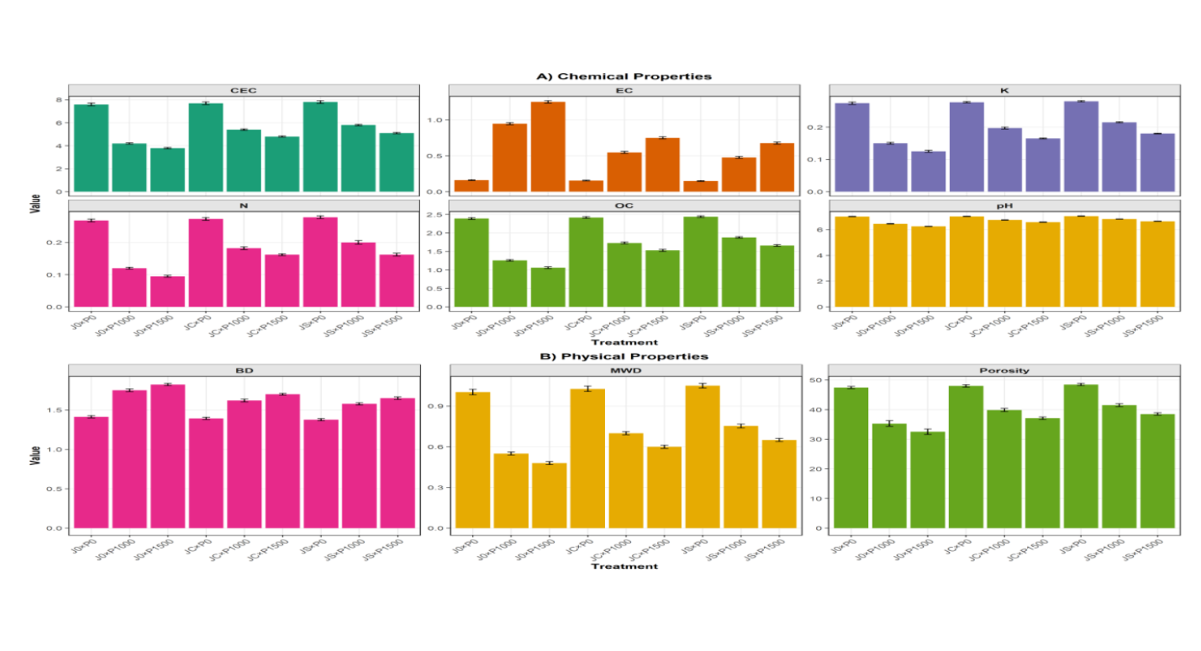
Figure 4: Soil Physicochemical Properties after Phytoremediation
Caption: Figure 4. Selected soil physicochemical properties under different treatments after 12 weeks: (a) Soil pH, (b) Organic Carbon (%), (c) Cation Exchange Capacity (cmol/kg), (d) Available Phosphorus (mg/kg). J0 = Unplanted Control, JC = Jatropha from Cutting, JS = Jatropha from Seed. Values are means ± standard error. Different letters indicate significant differences at p < 0.05 (Tukey's HSD).
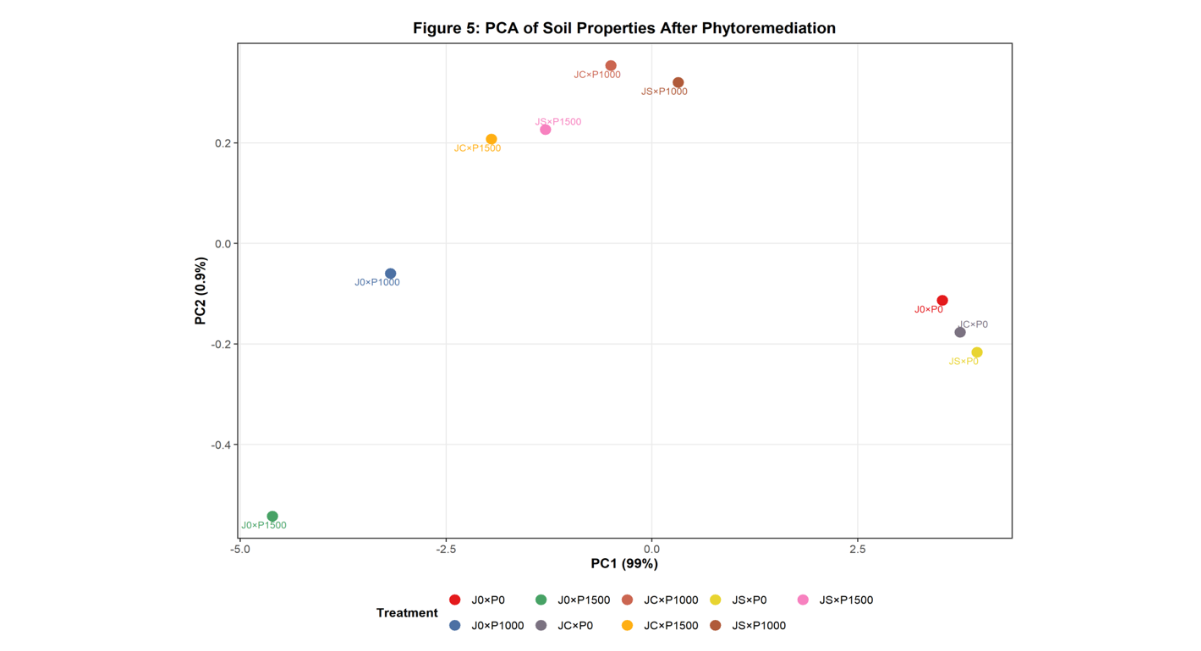
Figure 5: Principal Component Analysis (PCA) of Soil Properties
Caption: Figure 5. Principal Component Analysis biplot showing relationships among soil physicochemical properties and treatment groups. Vectors indicate direction and strength of variable influence on treatment separation.
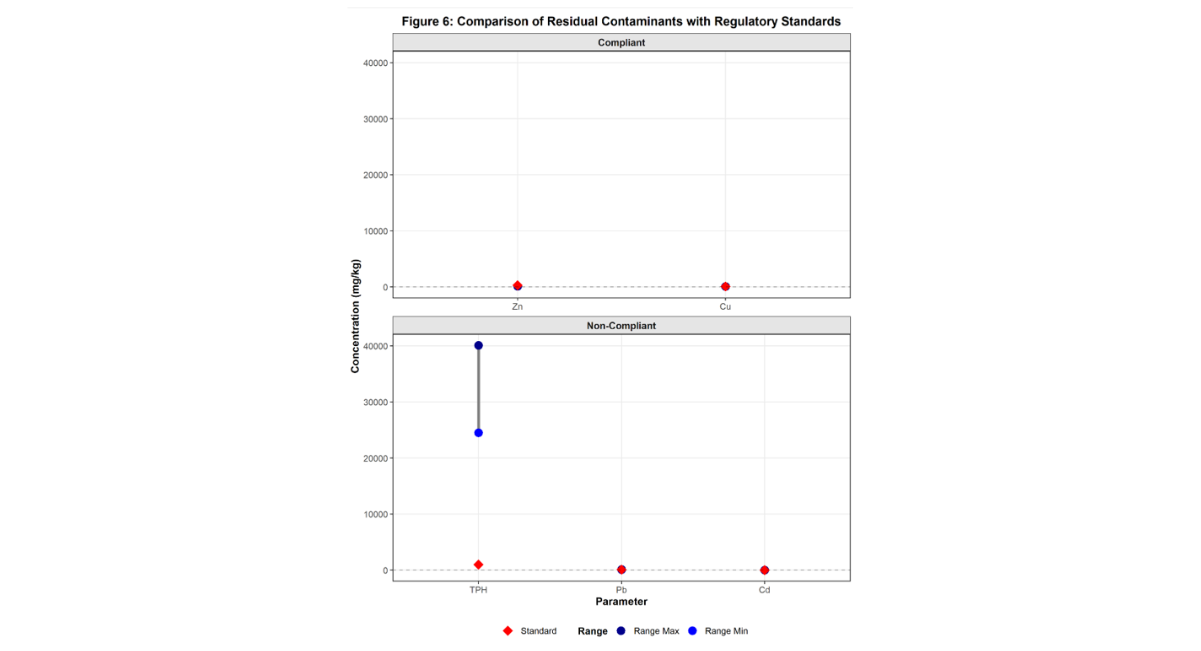
Comparison with Regulatory Standards
Residual contaminant concentrations were compared with national and international regulatory standards (Table 5).
|
Parameter |
Range (mg/kg) |
Standard (mg/kg) |
Compliance Status |
|
TPH |
24,530.5 – 40,104.0 |
1,000 (DPR, 2002) |
Non-Compliant |
|
Pb |
96.9 – 110.7 |
100 (FAO, 2020); 85 (WHO, 2020) |
Non-Compliant |
|
Cd |
3.20 – 3.79 |
3 (FAO, 2020); 0.8 (WHO, 2020) |
Non-Compliant |
|
Zn |
120.5 – 138.8 |
300 (FAO, 2020); 300 (WHO, 2020) |
Compliant |
|
Cu |
83.9 – 94.2 |
100 (FAO, 2020); 100 (WHO, 2020) |
Compliant |
Table 5: Comparison of Residual Contaminants with Regulatory Standards
All TPH values in vegetated soils remained well above the DPR ecological intervention limit of 1,000 mg/kg. Pb concentrations slightly exceeded FAO and WHO limits [47-49]. Cd concentrations exceeded all permissible limits. Zn and Cu remained within acceptable limits.
Soil fertility parameters (pH, OC, N, P, CEC) in remediated soils fell within FAO medium fertility ranges, indicating partial restoration [48]. However, bulk density (1.59–1.68 g/cm³) exceeded the FAO optimum (<1.60 g/cm³), and porosity (37.0–42.5%) was lower than the uncontaminated control (48.5%), suggesting slight structural deterioration.
DISCUSSION
Phytoremediation of Petroleum Hydrocarbons
The significant reduction in TPH concentration in vegetated treatments compared to unplanted controls confirms the effectiveness of Jatropha curcas in enhancing hydrocarbon degradation. The higher remediation efficiency achieved by seed-propagated plants (62.4–62.7%) compared to cutting-propagated plants (56.4–59.3%) can be attributed to differences in root architecture and rhizosphere dynamics. Seed-propagated Jatropha develops a deeper taproot system, which enhances soil aeration, increases the volume of rhizosphere soil, and supports a more diverse and active microbial community capable of hydrocarbon degradation [50-51]. Root exudates from seed-propagated plants likely provided carbon sources that stimulated hydrocarbon-degrading microbes, accelerating rhizodegradation [24,52]. In contrast, cutting-propagated plants, with shallower fibrous roots, had a more limited rhizosphere volume, reducing contact with deeper contaminants and limiting microbial stimulation. These findings align with Al-Hadith, who reported that propagation method significantly influences root development and pollutant uptake in Jatropha curcas [26]. Rao similarly found that seed-propagated plants exhibited greater root biomass and deeper penetration, enhancing their remediation potential [35]. Despite achieving substantial reductions (up to 62.7%), residual TPH concentrations (24,530–40,104 mg/kg) remained far above the DPR intervention limit of 1,000 mg/kg. This indicates that a single 12-week remediation cycle is insufficient for complete restoration of heavily contaminated soils [47]. Similar observations have been reported by Pilon-Smits and Prasad, who emphasized that phytoremediation efficiency depends on contamination load and treatment duration, with heavily polluted sites requiring multiple cropping cycles or integrated approaches [17,53]. When compared with regulatory standards, the residual TPH values in all vegetated soils remained well above the DPR ecological intervention limit of 1,000 mg/kg, indicating that extended remediation periods are necessary [47].
Heavy Metal Remediation
The differential reduction patterns among heavy metals reflect the specific uptake and accumulation mechanisms of Jatropha curcas. The significant reductions in Zn and Cu concentrations indicate effective phytoextraction of these essential micronutrients, which are required for plant metabolic processes including enzyme activation and photosynthesis, driving their active uptake and accumulation in plant tissues [22,52]. In contrast, Pb and Cd reductions were more limited, suggesting that Jatropha curcas primarily stabilizes these non-essential toxic metals in the rhizosphere (phytostabilization) rather than translocating them to harvestable tissues [23]. Ali explained that non-essential metals are often sequestered in root vacuoles or bound to cell walls, limiting their upward movement [19]. The superior performance of seed-propagated plants in reducing all metal concentrations further supports the importance of root architecture in metal uptake and stabilization. Deeper root systems provide greater surface area for metal adsorption and access to contaminants throughout the soil profile [25]. When compared with international regulatory standards, Zn concentrations (120.5–138.8 mg/kg) fell within FAO and USEPA permissible limits, and Cu concentrations (83.9–94.2 mg/kg) also complied with FAO and WHO standards [47,48,53]. However, Pb concentrations (96.9–110.7 mg/kg) exceeded FAO limit of 100 mg/kg and WHO limit of 85 mg/kg, while Cd concentrations (3.20–3.79 mg/kg) exceeded both FAO limit of 3 mg/kg and WHO limit of 0.8 mg/kg [48,49]. These findings indicate that while Jatropha curcas effectively manages micronutrient contaminants within acceptable timeframes, remediation of toxic metals to regulatory standards requires extended periods or integrated approaches.
Soil Physicochemical Restoration
The improvement in soil physicochemical properties under Jatropha curcas vegetation demonstrates the plant's capacity to restore soil health beyond simple contaminant removal. The increase in soil pH from acidic levels (6.25) in contaminated controls to near-neutral values (6.65–6.82) in vegetated treatments can be attributed to the release of basic cations (Ca²⁺, Mg²⁺, K⁺) through root activity and microbial decomposition of organic residues [50-51]. This pH neutralization is critical for reducing metal bioavailability and creating favorable conditions for microbial activity and nutrient cycling. The increase in organic carbon (OC) in vegetated soils (1.53–1.88%) compared to unplanted contaminated controls (1.06–1.26%) reflects continuous addition of root exudates, senesced root tissues, and leaf litter, which serve as substrates for microbial growth and contribute to soil organic matter buildup [54-55]. Higher OC in seed-propagated treatments suggests greater root biomass and exudation compared to cuttings. The improvement in CEC (4.80–5.80 cmol/kg) correlates strongly with increased OC, as organic matter contributes significantly to the soil's exchange capacity [56-57]. This enhancement in nutrient retention capacity is essential for supporting subsequent vegetation and agricultural productivity on remediated sites. Increased available phosphorus (15.5–18.8 mg/kg) in vegetated treatments likely results from organic acid and phosphatase release by Jatropha roots and associated microbes, which solubilize bound phosphorus [58-59]. Higher values in seed-propagated treatments again indicate greater rhizosphere activity. The reduction in bulk density (1.59–1.68 g/cm³) and increase in porosity (37.0–42.5%) in vegetated soils compared to unplanted contaminated controls (1.75–1.82 g/cm³; 32.5–35.3%) reflect improved soil structure through root penetration, aggregate formation, and organic matter incorporation [29]. However, values had not fully recovered to uncontaminated control levels (1.39 g/cm³; 48.5%), indicating that structural restoration requires longer timeframes than chemical recovery. Soil fertility parameters in remediated soils fell within FAO (2020) medium fertility ranges, indicating partial restoration. However, bulk density (1.59–1.68 g/cm³) exceeded the FAO optimum (<1.60 g/cm³), and porosity (37.0–42.5%) was lower than the uncontaminated control (48.5%), suggesting slight structural deterioration that requires continued management.
Comparison with Regulatory Standards and Implications
The failure of residual TPH, Pb, and Cd concentrations to meet regulatory standards after 12 weeks has important implications for phytoremediation practice in the Sudan Savannah. While Jatropha curcas demonstrated significant remediation capacity, the high initial contamination levels (70,833–106,250 mg/kg TPH) exceeded the typical range for which phytoremediation alone is recommended. Prasad noted that phytoremediation is most effective for low-to-moderate contamination levels, with heavily polluted sites requiring integrated approaches. However the compliance of Zn and Cu with international standards suggests that Jatropha curcas can effectively manage micronutrient contaminants within acceptable timeframes. This differential performance based on metal type should inform site-specific remediation planning, with expectations adjusted for different contaminant classes. The partial restoration of soil fertility parameters to FAO (2020) medium ranges indicates that phytoremediation with Jatropha curcas can simultaneously achieve contaminant reduction and soil quality improvement. This dual benefit enhances the economic and environmental viability of the approach, particularly in resource-limited settings where land rehabilitation must support eventual agricultural use [29-30].
CONCLUSION
This study demonstrates that Jatropha curcas possesses significant potential for the phytoremediation of petroleum hydrocarbon and heavy metal contaminated soils in the Sudan Savannah ecological zone of Nigeria. The findings conclusively show that propagation method substantially influences remediation efficiency, with seed-propagated Jatropha curcas achieving superior performance compared to cutting-propagated plants. Seed-propagated plants recorded the highest TPH removal efficiencies of 62.4% and 62.7% at 1000 mL and 1500 mL contamination levels respectively, outperforming cutting-propagated plants which achieved 56.4% and 59.3% removal. This enhanced performance is attributed to the deeper taproot system of seed-grown plants, which increases rhizosphere volume, stimulates greater microbial activity, and provides more extensive contact with contaminants throughout the soil profile. The research further establishes that Jatropha curcas exhibits differential remediation capacity across contaminant types. The plant effectively remediated essential micronutrients zinc and copper, but remediation of toxic metals (lead and cadmium) and total petroleum hydrocarbons to regulatory standards requires extended timeframes, as residual concentrations remained above regulatory limits despite significant reductions. Importantly, this study reveals that phytoremediation with Jatropha curcas delivers dual benefits beyond contaminant removal. Significant improvements in soil physicochemical properties were observed across all vegetated treatments, with seed-propagated plants showing superior restoration capacity. Soil pH increased, organic carbon content improved by up to 23%, and cation exchange capacity, available phosphorus, and total nitrogen all showed substantial increases. Concurrently, bulk density decreased and porosity improved, indicating partial recovery of soil physical structure. These improvements demonstrate that Jatropha curcas not only removes or stabilizes contaminants but actively restores soil health and fertility, creating conditions suitable for subsequent agricultural use or ecological recovery. The dual economic and environmental benefits contaminant remediation alongside potential biofuel production enhance the viability of phytoremediation approaches in resource-limited settings where conventional cleanup methods are prohibitively expensive.
RECOMMENDATIONS
For practical application, seed propagation should be adopted as the preferred method for large-scale phytoremediation projects in the Sudan Savannah and similar semi-arid environments. The superior performance of seed-propagated plants in both contaminant removal and soil restoration justifies the additional time required for initial establishment compared to vegetative propagation. For sites with high contamination levels comparable to those in this study (70,000–106,000 mg/kg TPH), remediation duration should be extended beyond 12 weeks, or multiple cropping cycles should be planned to achieve regulatory compliance, particularly for TPH, lead, and cadmium. The integration of Jatropha curcas phytoremediation with complementary approaches including bioaugmentation with hydrocarbon-degrading microbial consortia, organic amendments such as biochar or compost, and mycorrhizal inoculation is strongly recommended to accelerate contaminant removal and enhance soil structural recovery. From a policy perspective, government agencies and environmental management bodies should incorporate Jatropha curcas into national oil-spill response frameworks and land rehabilitation programs, particularly for semi-arid regions where the plant's drought tolerance provides distinct advantages. Funding mechanisms and technical training programs should be established to support community-led phytoremediation initiatives, enabling local participation in land restoration while generating potential income from biofuel production. For future research, long-term field studies spanning multiple growing seasons are urgently needed to evaluate the full remediation potential of Jatropha curcas under actual field conditions in the Sudan Savannah, accounting for seasonal variations, natural precipitation patterns, and real-world contaminant heterogeneity. Detailed quantification of hydrocarbon and metal accumulation in plant tissues roots, stems, leaves, and seeds is essential to assess potential risks associated with biofuel utilization and to develop appropriate harvest, disposal, and processing protocols that prevent secondary contamination. Investigation of microbial community dynamics in the rhizosphere of seed-propagated versus cutting-propagated plants would elucidate the mechanisms underlying differential remediation efficiency, potentially identifying key microbial taxa or enzymatic processes that could be enhanced through management practices.
ACKNOWLEDGMENTS
The authors gratefully acknowledge the Department of Soil Science and Forestry, Aliko Dangote University of Science and Technology, Wudil, for providing nursery facilities for this research. We thank the Federal University Duste for laboratory analysis support. Special appreciation is extended to Mubarak Mustapha for technical assistance during soil sampling and laboratory analyses.
FUNDING
This research received no external funding.
AUTHOR CONTRIBUTIONS
- Mus'ab Bashir Yusuf: Conceptualization, methodology, investigation, formal analysis, writing original draft, writing review and editing.
- Abdulkadir Sani: Supervision, methodology, writing review and editing.
- Onokebhagbe Odiamehi Victor: Data curation, validation, writing review and editing.
- Zainab Hassan: Technical assistance during soil sampling and laboratory analyses
CONFLICTS OF INTEREST
The authors declare no conflicts of interest.
DATA AVAILABILITY STATEMENT
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
REFERENCES
- UNEP. (2022). Global assessment of soil pollution: Report. United Nations Environment Programme. [Crossref]
- FAO. (2023). The state of food security and nutrition in the world 2023. Food and Agriculture Organization of the United Nations. [Crossref]
- Atagana HI. Bioremediation of co-contamination of crude oil and heavy metals in soil by phytoremediation using Chromolaena odorata (L) King & HE Robinson. Water, Air, & Soil Pollution. 2011;215(1):261-71. [Crossref] [Google Scholar]
- Bamgbose, I, Anderson, C. W, Rutter A. (2021). Phytostabilization of hydrocarbon-contaminated soils using Jatropha curcas. Environmental Science and Pollution Research, 28(10), 12345-12356.
- Chibuike GU, Obiora SC. Heavy metal polluted soils: effect on plants and bioremediation methods. Applied and environmental soil science. 2014;2014(1):752708. [Crossref] [Google Scholar]
- Wang, Y, Li, F, Ruan X, Song J (2022). Impact of petroleum hydrocarbons on soil microbial community and function. Environmental Pollution, 292, 118448. [Crossref]
- Alloway BJ. Trace metals and metalloids in soils and their bioavailability. Heavy Metals in Soils. 2013;22:4-18. [Google Scholar]
- Ali H, Khan E, Ilahi I. Environmental chemistry and ecotoxicology of hazardous heavy metals: environmental persistence, toxicity, and bioaccumulation. Journal of chemistry. 2019;2019(1):6730305. [Crossref] [Google Scholar]
- Ijah, U. J. J., Ayangeen, A. S., & Okafor, B. Microbiological and physicochemical characteristics of soil contaminated with used petroleum products in Benue State, Nigeria. International Journal of Environmental Science and Technology, 11(4), 1081-1090.
- Olokeogun OS, Kumar R. Indicator-based vulnerability assessment of riparian zones in Nigeria’s Ibadan region due to urban settlement pressure. Environ Chall 7: 100501. 2022. [Crossref] [Google Scholar]
- Adelekan BA, Abegunde KD. Heavy metals contamination of soil and groundwater at automobile mechanic villages in Ibadan, Nigeria. International Journal of the physical Sciences. 2011;6(5):1045-58. [Google Scholar]
- Emolade MO, Anozie JA, Adinkwu OM, Isoje AO, Aphiar AE, Okere OL, et al. Assessment of Heavy Metal Contamination in Soils Around Auto-Mechanic Workshops in Ozoro, Delta State, Nigeria. Advances in Research on Teaching. 2025;26(3):342-50. [Crossref] [Google Scholar]
- Gambo A, Fullen MA, Baldwin TC. Characterization and classification of soils on a toposequence around Dutse Jigawa State, Nigeria. Discover Soil. 2025;2(1):97. [Crossref] [Google Scholar]
- Ansari AA, Gill SS, Gill R, Lanza GR, Newman L, editors. Phytoremediation: management of environmental contaminants, volume 1. Springer; 2014. [Crossref] [Google Scholar]
- Salt DE, Smith RD, Raskin I. Phytoremediation. Annual review of plant biology. 1998;49(1):643-68.. [Crossref] [Google Scholar] [PubMed]
- Chaney RL, Malik M, Li YM, Brown SL, Brewer EP, Angle JS, Baker AJ. Phytoremediation of soil metals. Current opinion in Biotechnology. 1997;8(3):279-84. [Crossref] [Google Scholar] [PubMed]
- Pilon-Smith E. Phytoremediation: annual review plant biology. [Crossref] [Google Scholar] [PubMed]
- Tangahu BV, Sheikh Abdullah SR, Basri H, Idris M, Anuar N, Mukhlisin M. A review on heavy metals (As, Pb, and Hg) uptake by plants through phytoremediation. International journal of chemical engineering.2011(1):939161. [Crossref] [Google Scholar]
- Ali H, Khan E, Sajad MA. Phytoremediation of heavy metals—concepts and applications. Chemosphere. 2013;91(7):869-81. [Crossref] [Google Scholar] [PubMed]
- Pandey VC, Singh K, Singh JS, Kumar A, Singh B, Singh RP. Jatropha curcas: A potential biofuel plant for sustainable environmental development. Renewable and sustainable energy reviews. 2012;16(5):2870-83. [Crossref] [Google Scholar]
- Puthur JT, Dhankher OP. Bioenergy Crops. Boca Raton, FL, USA: CRC Press; 2022. [Google Scholar]
- Álvarez-Mateos P, Alés-Álvarez FJ, García-Martín JF. Phytoremediation of highly contaminated mining soils by Jatropha curcas L. and production of catalytic carbons from the generated biomass. Journal of environmental management. 2019;231:886-95. [Crossref] [Google Scholar] [PubMed]
- Juwarkar AA, Singh SK, Mudhoo A. A comprehensive overview of elements in bioremediation. Reviews in Environmental Science and bio/technology. 2010;9(3):215-88. [Crossref] [Google Scholar]
- Mametja NM, Ramadwa TE, Managa M, Masebe TM. Recent advances and developments in bacterial endophyte identification and application: A 20-Year landscape review. Plants. 2025;14(16):2506. [Crossref] [Google Scholar] [PubMed]
- Donatus, F.N, Akogwu, R. D. Phytoremediation of Petroleum Hydrocarbons Using Jatropha curcas in Soils Contaminated with Spent Engine Oil. International Journal of Research and Scientific Innovation, Volume VIII, Issue IV, 2021.
- Chang FC, Ko CH, Tsai MJ, Wang YN, Chung CY. Phytoremediation of heavy metal contaminated soil by Jatropha curcas. Ecotoxicology. 2014;23(10):1969-78. [Crossref] [Google Scholar] [PubMed]
- Edegbai BO, Anoliefo GO. Growth and development of Bitter leaf (Vernonia amygdalina Del.) in soils treated with mixture of cadmium and lead. Journal of Applied Science & Environmental Management. 2019;23(4):835-41. [Crossref] [Google Scholar]
- Agamuthu P, Abioye OP, Aziz AA. Phytoremediation of soil contaminated with used lubricating oil using Jatropha curcas. Journal of hazardous materials. 2010 Jul 15;179(1-3):891-4. [Crossref] [Google Scholar] [PubMed]
- Bashir MA. Phytoremediation of heavy metals contaminated agricultural soil planted to Jatropha curcas. International Journal of Energy and Environmental Research Vol. 10, No. 3, pp., 14-32, 2022 Print ISSN-2055-0197 (Print), Online ISSN 2055-0200. 2022. [Crossref] [Google Scholar]
- Awotedu, O. L., & Ogunbamowo, P. O. (2019). Comparative heavy metal uptake and phytoremediation potential of three Jatropha species. Environment & Ecosystem Science, 3(2), 26–30.
- (2021). World reference base for soil resources 2020. Food and Agriculture Organization of the United Nations.
- Mustapha AA, Abdu N, Oyinlola EY, Nuhu AA, Dawaki MU. Distribution of aluminium, iron, calcium, magnesium and manganese in the soils of Savannah Region of Northern Nigeria. Journal of Agricultural Economics, Environment and Social Sciences. 2020;6(1):32-9. [Google Scholar]
- Ayotamuno JM, Kogbara RB, Egwuenum PN. Comparison of corn and elephant grass in the phytoremediation of a petroleum-hydrocarbon-contaminated agricultural soil in Port Harcourt, Nigeria. Journal of Food Agriculture and Environment. 2006;4(3/4):218. [Google Scholar]
- Abosede EE . Effect of crude oil pollution on some soil physical properties. Journal of agriculture and veterinary science. 2013;6(3):14-7. [Crossref] [Google Scholar]
- Behera SK, Srivastava P, Tripathi R, Singh JP, Singh N. Evaluation of plant performance of Jatropha curcas L. under different agro-practices for optimizing biomass–a case study. Biomass and bioenergy. 2010;34(1):30-41. [Crossref] [Google Scholar]
- Chen G, Li N, Cheng Z, editors. Solid Waste-based Materials for Environmental Remediation. New York: CRC Press; 2024. [Google Scholar]
- Nero BF. Phytoremediation of petroleum hydrocarbon-contaminated soils with two plant species: Jatropha curcas and Vetiveria zizanioides at Ghana Manganese Company Ltd. International Journal of Phytoremediation. 2021;23(2):171-80. [Crossref] [Google Scholar] [PubMed]
- Nelson DW, Sommers LE. Total carbon, organic carbon, and organic matter. Methods of soil analysis: Part 2 chemical and microbiological properties. 1982;9:539-79. [Crossref] [Google Scholar]
- (2007). Method 8015B: Nonhalogenated organics by gas chromatography. United States Environmental Protection Agency.
- (2019). Official methods of analysis (21st ed.). Association of Official Analytical Chemists.
- Estefan G, Sommer R, Ryan J. Methods of soil, plant, and water analysis: a manual for the West Asia and North Africa region. [Google Scholar]
- Walkley A. A critical examination of a rapid method for determining organic carbon in soils—effect of variations in digestion conditions and of inorganic soil constituents. Soil science. 1947;63(4):251-64. [Crossref] [Google Scholar]
- Bremner JM, Mulvaney CS. Nitrogen—total. Methods of soil analysis: part 2 chemical and microbiological properties. 1982;9:595-624. [Crossref] [Google Scholar]
- Bray RH, Kurtz LT. Determination of total, organic, and available forms of phosphorus in soils. Soil science. 1945;59(1):39-46. [Crossref] [Google Scholar]
- Chapman HD. Cation‐exchange capacity. Methods of soil analysis: Part 2 Chemical and microbiological properties. 1965;9:891-901. [Crossref] [Google Scholar]
- Gomez KA, Gomez AA. Statistical procedures for agricultural research. John wiley & sons; 1984. [Google Scholar]
- DPR (Department of Petroleum Resources). (2002). Environmental Guidelines and Standards for the Petroleum Industry in Nigeria (EGASPIN). Lagos, Nigeria: Department of Petroleum Resources.
- Fao F. Agriculture organization of the United Nations. 2012. FAO statistical yearbook. 2012. [Google Scholar]
- World Health Organization. Guidelines for the safe use of wastewater, excreta and greywater. World Health Organization; 2006. [Crossref] [Google Scholar]
- Eze, M. O., Thiel, V., Hose, G. C., George, S. C., & Daniel, R. Enhancing rhizoremediation of petroleum hydrocarbons through bioaugmentation with a plant growth-promoting bacterial consortium. Chemosphere, 289, 133143. 2022. [Crossref] [PubMed]
- Ogunwole JO, Chaudhary DR, Ghosh A, Daudu CK, Chikara J, Patolia JS. Contribution of Jatropha curcas to soil quality improvement in a degraded Indian entisol. Acta Agriculturae Scandinavica Section B–Soil and Plant Science. 2008;58(3):245-51. [Crossref] [Google Scholar]
- Ghosh M, Singh SP. A review on phytoremediation of heavy metals and utilization of it’s by products. Asian J Energy Environ. 2005;6(4):18. [Crossref] [Google Scholar]
- Prasad R, editor. Phytoremediation for environmental sustainability. Singapore: Springer; 2021. [Crossref] [Google Scholar]
- Lal R. Soil organic matter content and crop yield. Journal of Soil and Water Conservation. 2020;75(2):27A-32A. [Crossref] [Google Scholar]
- Saravanan N, Puhan S, Nagarajan G, Vedaraman N. An experimental comparison of transesterification process with different alcohols using acid catalysts. Biomass and bioenergy. 2010;34(7):999-1005. [Crossref] [Google Scholar]
- Brady NC, Weil RR, Weil RR. The nature and properties of soils. Upper Saddle River, NJ: Prentice Hall; 2008. [Crossref] [Google Scholar]
- Havlin JL, Tisdale SL, Nelson WL, Beaton JD. Soil fertility and fertilizers. Pearson Education India; 2016. [Crossref] [Google Scholar]
- Khan S, El-Latif Hesham A, Qiao M, Rehman S, He JZ. Effects of Cd and Pb on soil microbial community structure and activities. Environ Sci Pollut Res Int. 2010;17(2):288-96. [Crossref] [Google Scholar] [PubMed]
- Egobueze FE, Ayotamuno JM, Iwegbue CM, Eze C, Okparanma RN. Effects of organic amendment on some soil physicochemical characteristics and vegetative properties of Zea mays in wetland soils of the Niger Delta impacted with crude oil. International journal of Recycling of organic waste in Agriculture. 2019: 423-35. [Crossref] [Google Scholar]
Article Processing Timeline
| 2-5 Days | Initial Quality & Plagiarism Check |
| 25-35 Days |
Peer Review Feedback |
| 45-60 Days | Total article processing time |
Journal Flyer