ABSTRACT
Background
Vasovagal Syncope (VVS) shares the autonomic system effect and commonality with smoking, raising the question of this study about the possibility of tending to smoke among medical students to prevent the VVS.
Methods
A cross-sectional study was conducted using a self-administered online questionnaire distributed to Jordanian clinical medical students, interns, and junior residents. From 525 initial responses, 456 participants met the inclusion criteria. VVS was defined based on the clinical criteria from the self-responded questionnaires.
Keywords: Syncope; Vaso Vagal Syncope; Smoking; Pathogenesis; Autonomic Dysfunction; Smoking Effects; Cross-Sectional Study
INTRODUCTION
Syncope is a self-limited, transient loss of consciousness [1]. Vasovagal syncope (VVS), also known previously as neurocardiogenic syncope, is the most common type, with 60-70% of all syncopes [2].The consensus mechanism of VVS goes to the increased parasympathetic tone and sympathetic activity withdrawal causing the well know parasympathetic sequences of bradycardia, vasodilatation, and decreased peripheral resistance leading to cerebral hypoperfusion [3,4]. The triggers remain questionable however answers go towards the defense mechanism of the higher centers or ventricular mechanoreceptors' paradoxical firing against the brain stem shutting down [5,6]. However, other sources debate it as a psychological issue, like anxiety that activates sympathetic stimulation [7]. Also, Family history and sex of the subject are important predictors of vasovagal syncope in offspring [8]. The prevalence of syncope for a lifetime of 70 years is 42%, with an annual rate of 6% for a lifetime of 70 years. Moreover, the incidence ranges from 18.1 to 39.7 per 1,000 patients [9].
Medical students experience significant stress-related morbidity, documented with nonrefreshing sleep, moderate-severe sleep disturbances, and insomnia (40.6%, 31.1%, and 38.4%, respectively). In addition to, anxiety (41.3%), and depression (44.6%)[10]. This population demonstrates concerning lifestyle habits, including a 49.5% smoking prevalence in Georgian medical students [11]. Vasovagal syncope (VVS) affects this group substantially, with a 2009 study of 630 clinical students reporting 12% prevalence (77 cases), showing marked female predominance (88%) and a mean age of 23 years (range 20-45).Among the students affected, 22% were graduate students while 78% were undergraduates. Of these, 57% (44 out of 77) intended to pursue surgical careers, and 9% (7 out of 77) reported feeling discouraged from surgery due to their episodes. The most common triggers identified were exposure to hot environments (79%, n=61) and prolonged periods of standing (73%, n=56). surgical masks (47%, n=36), and diathermy smell (23%, n=18), while preventive measures included pre-theater nutrition (61%, n=47) and leg movement (18%, n=14) [12].
The pathophysiology involves NO-mediated vasodilation during VVS episodes (220% urinary cGMP increase versus 67% decrease in controls)[4], while smoking appears to modulate autonomic responses by reducing baseline vagal tone while offering cerebral perfusion protection during arrhythmias trough enhancing sympathetic vasoconstriction[13]. Familial studies of 62 medical students and 228 relatives, with a median first faint age of 14 years reveal important predispositions, demonstrating 32% VVS prevalence. Transmission patterns show stronger maternal inheritance (affecting both sexes) while paternal inheritance significantly impacts only sons.
Despite this background, the specific relationship between smoking and VVS risk remains unexplored in medical trainees.
This study aimed to:
1) determine the prevalence of VVS among Jordanian clinical medical students, interns, and junior residents;
2) test the hypothesis that smoking is associated with a lower prevalence of VVS due to its sympathomimetic effects;
3) identify other associated lifestyle and familial factors.
METHODS
Study Design and Sample Size
This cross-sectional study explored the relationship between episodes of syncope and the likelihood of smoking among clinical medical students, interns, and junior residents—a group especially susceptible to the pressures associated with medical training. Existing literature indicates stress prevalence of 31.7% among medical students and 73.0% among interns [14,15].
Sample size calculation was performed using G*Power 3.1 and EpiInfo software. For a Chi-square test with a 95% confidence level, 5% margin of error, and a 50% effect size, a minimum of 385 participants was required. We collected 525 responses. After applying exclusion criteria (cardiovascular diseases, anemia, diabetes mellitus, seizures, other syncope types, or incomplete questionnaires), the final analytical sample comprised 456 participants. Data were collected through an online self-administered English questionnaire using convenience sampling. The data were collected from 10th January 2025 to 16th Feb 2025.
Data Collection Instrument
The survey consisted of 30 items and was structured into four main domains: sociodemographic data (including age, sex, and academic level), medical history (covering cardiac, neurological, and other relevant comorbidities), lifestyle-related factors (such as smoking behavior, physical activity, and family history), and features of vasovagal syncope (including triggers, symptoms, duration, and relieving factors). The questionnaire incorporated a variety of formats, including yes/no questions, multiple-choice items, checklists, and a limited number of open-ended responses.
Questionnaire Development and Validation
Given the lack of existing validated tools that simultaneously evaluate vasovagal syncope and smoking behavior, a comprehensive questionnaire was designed by adapting relevant items from previously validated instruments identified through searches of databases such as PubMed and Scopus. With authors' permission incorporating established syncope assessment tools (e.g., Calgary Syncope Score) [9,16,17]. The questionnaire underwent rigorous validation: expert review by impartial analysts and a consultant cardiologist to: eliminate double-barreled, ambiguous, or misleading questions, ensure scientific accuracy, and establish content and face validity. Then, pilot testing (n>30) to evaluate was conducted to evaluate readability and comprehension, construct validity, criterion validity, and internal consistency (reliability).
Operational Definitions
Vasovagal syncope (VVS): Was defined operationally based on self-report of a transient loss of consciousness and/or typical pre-syncopal symptoms in response to common VVS triggers (e.g., fear, pain, prolonged standing, sight of blood) as per the questionnaire items with free medical history.
Smoking status: Participants were classified as 'smokers' if they reported current use of cigarettes, vapes, or shisha.
Statistical Analysis
All statistical analyses were conducted using SPSS version 26.0 (IBM). Participants older than 30 years, as well as those with cardiovascular conditions (including arrhythmias, orthostatic hypotension, first-degree atrioventricular block, hypertension, or valvular heart disease), neurological disorders (such as epilepsy or other seizure conditions), anemia, or diabetes were excluded. Individuals with primary headache disorders were retained. After applying these criteria, a total of 456 participants were included in the final analysis, and any records containing missing data were omitted.
Descriptive statistics were calculated for all demographic and behavioral variables. Associations between these variables and vasovagal syncope were assessed using Pearson’s chi-square (χ²) test.
A full‑model(forced entry) binary logistic regression was then fitted with vasovagal syncope (0 = did not experience vasovagal syncope, 1 = experience vasovagal syncope) as the dependent variable, and age, weight, nightly sleep hours, smoking status, sex, caffeine use, gum consumption, family history of vasovagal syncope, number and type of smoking products, perceived stress, changes in smoking habits and physical‑activity frequency entered simultaneously as predictors, Variable inclusion was based on theoretical relevance and prior findings in the literature.. Results are reported as B coefficients, standard errors, Wald χ² statistics, p‑values, adjusted odds ratios (Exp B) and their 95 % confidence intervals. Logistic‑regression assumptions were checked: model fit was adequate (Pearson χ² = 459.8, df = 425, p = 0.12; deviance χ² = 384.8, df = 425, p = 0.92) and all variance‑inflation factors were < 2.5, indicating no multicollinearity.
ETHICAL CONSIDERATIONS
The study received ethical approval from the University of Jordan Institutional Review Board (IRB Code: 1667/2025/67). Key ethical provisions included: obtaining informed consent from all participants, ensuring participant anonymity and data confidentiality, guaranteeing voluntary participation with the right to withdraw, and emphasizing that there is no provision of short-term benefits or rewards.
RESULTS
The study analyzed data from 525 initially collected responses, with 456 participants meeting inclusion criteria after excluding those with anemia, diabetes, or seizure history. The final sample comprised 56.8% females and 43.2% males, with a mean age of 23.76 years (SD=1.97). Most participants were fourth-year medical students and interns, predominantly single (72.3%) and with normal BMI (58.1%) (Table 1). A statistically significant association was observed between gender and vasovagal syncope (VVS) occurrence (p=0.008), while BMI showed no significant relationship (p=0.393). The overall prevalence of self-reported VVS was 20.6% (n=94). Of the 121 smokers (26.5%), 22 (18.2%) reported VVS, compared to 72 of 335 nonsmokers (21.5%). No significant association was found between smoking status and VVS (χ²=0.112, p=0.738). In multivariable analysis, significant independent predictors of VVS were female gender (Adjusted Odds Ratio [AOR]=1.92, p=0.012), positive family history (AOR=8.66, p<0.001), and engaging in weekly physical activity (AOR=0.30, p=0.004).
Smoking characteristics revealed that 26.5% (n=121) of participants were current smokers, among whom 18.2% (n=22) reported VVS episodes. Comparatively, 21.5% (n=72) of non-smokers (n=335) experienced VVS. Multiple logistic regression analysis demonstrated no significant association between smoking status and VVS (Wald χ²=1.729, p=0.188), though vaping showed borderline statistical significance (p=0.056) that may warrant further investigation with larger samples (Table 2). Cross-tabulation of VVS cases by smoking status revealed no meaningful distributional differences (Pearson χ²=0.112, p=0.738), as illustrated in Figure 1.
Among the 94 confirmed VVS cases (population prevalence=20.6%), only 8.5% (n=8) had received a formal medical diagnosis, including 2 cases among smokers (Table 3).
To examine the effect of smoking status on the number of VVS episodes among the 94 students who had experienced syncope, a Poisson model was first estimated, but showed substantial over‑dispersion (Pearson χ²/df = 5.17); consequently, a negative‑binomial model was applied. The negative‑binomial model demonstrated good fit (Pearson χ²/df = 0.983). In that model, non‑smokers exhibited a 33 % lower episode rate than smokers (Exp B = 0.670, 95 % CI 0.408–1.100), although the difference was not statistically significant (p = 0.113).
Adjusted logistic regression controlling for gender, age, and comorbidities identified family history as the strongest predictor of VVS (p<0.001), with affected individuals demonstrating 8.7-fold increased odds (OR=8.658). Of the 42 participants reporting familial VVS, nearly half (47.6%, n=20) experienced personal episodes. Physical activity patterns showed a complex relationship, with weekly and occasional exercisers exhibiting 70.5% and 54.3% reduced VVS odds, respectively (p=0.004 and p=0.029), while extreme exercise patterns showed no association (p=0.581) (Table 4).
No statistically significant relationships were identified between VVS and sleep duration (OR = 0.873 per hour, p = 0.107), caffeine intake (OR = 0.855, p = 0.639), or gum-chewing behavior (OR = 1.175, p = 0.548). However, notable trends suggested potential protective effects of increased sleep (12.7% lower odds/hour) and caffeine abstinence (14.5% reduced odds), while non-gum chewers showed 17.5% elevated odds. Stress analysis proved inconclusive due to the absence of zero-stress respondents, though non-significant trends suggested higher stress levels might correlate with reduced VVS occurrence (Level 1 vs Level 3 stress: OR=8.567, p=0.144; Level 2 vs Level 3: OR=5.249, p=0.249) (Table 4).
|
Variable |
Category |
Count |
Percentage |
|
Gender |
Females |
259 |
56.80% |
|
Males |
197 |
43.20% |
|
|
Medical Year |
4th year |
111 |
24.30% |
|
5th year |
41 |
9.00% |
|
|
6th year |
82 |
18.00% |
|
|
Internship |
163 |
35.70% |
|
|
Junior residency |
59 |
12.90% |
|
|
Nationality |
Jordanian |
434 |
95.20% |
|
Non-Jordanian |
22 |
4.80% |
|
|
Training Hospital |
Jordan University Hospital |
118 |
25.90% |
|
Al-Basheer |
36 |
7.90% |
|
|
King-Abdullah University Hospital (KAUH) |
46 |
10.10% |
|
|
King Hussein Cancer Center (KHCC) |
6 |
1.30% |
|
|
King Hussein Medical Center (KHMC) |
33 |
7.20% |
|
|
Al-Karak Hospital |
124 |
27.20% |
|
|
Prince Hamzeh Hospital |
39 |
8.60% |
|
|
Private hospitals |
54 |
11.80% |
|
|
BMI |
< 18.5 |
22 |
4.80% |
|
18.5 – 24.9 |
230 |
50.40% |
|
|
25 – 29.9 |
160 |
35.10% |
|
|
30 – 34.9 |
36 |
7.90% |
|
|
35 – 39.9 |
6 |
1.30% |
|
|
≥ 40 |
2 |
0.40% |
|
|
Marital Status |
Single |
432 |
94.70% |
|
Engaged |
13 |
2.90% |
|
|
Married |
11 |
2.40% |
Table 1: Sociodemographic characteristics of the participants
|
Smoking Type / Habit |
Count (% Yes) |
p-Value for Syncope |
|
Cigarettes (1 = smoke) |
62 (13.6%) |
0.135 |
|
Vapes (1 = vape) |
63 (13.8%) |
0.056 |
|
Shisha (1 = hookah) |
52 (11.4%) |
0.273 |
|
No Change in Habit |
44 (9.6%) |
- |
|
Decreased Smoking |
29 (6.4%) |
0.273 |
|
Increased Smoking |
45 (9.9%) |
0.525 |
Table 2: Descriptive table for smoking types and habits among the participants
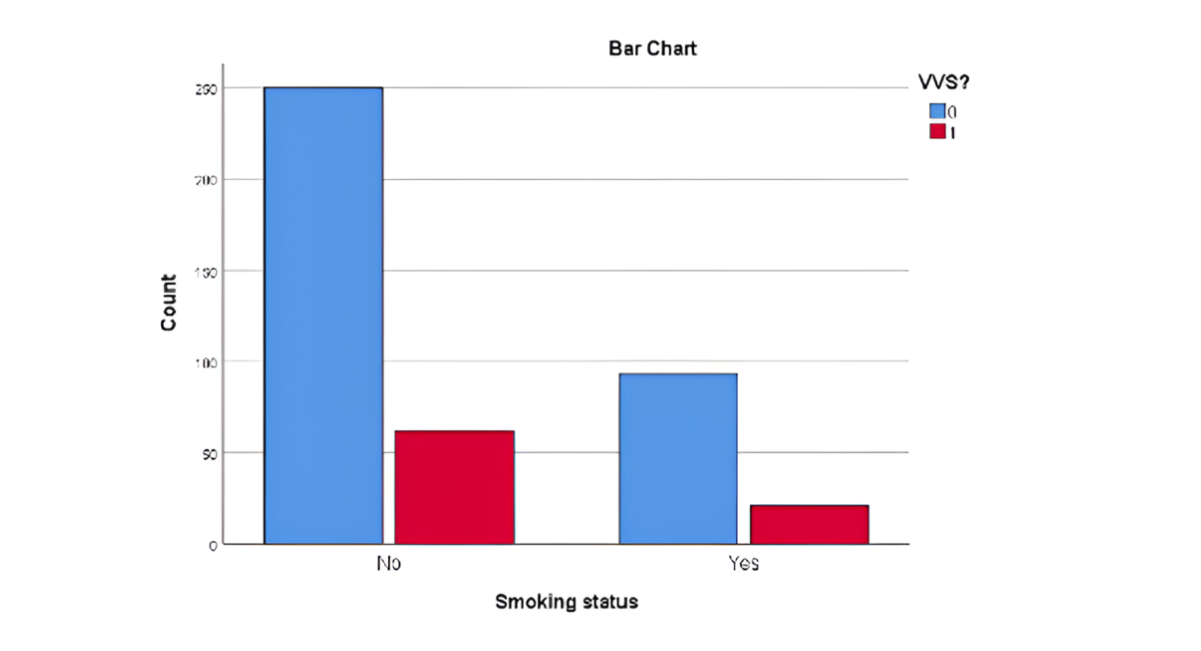
Figure 1: Clustered bar count of VVS by smoking status
Proportion Test Results
- Chi-square (Pearson): 0.112
- p-value from chi-square: 0.738
No significant association was found between smoking status and VVS. Further investigation in larger samples may still be warranted to confirm the trend.
|
S.no |
Characteristic |
Category |
Count (%) |
|
1 |
Clinically Diagnosed |
Yes |
8 (1.8%) |
|
No |
448 (98.2%) |
||
|
2 |
Number of VVS Attacks in a whole life |
0 |
362 |
|
-79.40% |
|||
|
1–5 |
78 (17.1%) |
||
|
5–10 |
12 (2.6%) |
||
|
10–15 |
1 (0.2%) |
||
|
15–20 |
2 (0.4%) |
||
|
>20 |
1 (0.2%) |
||
|
3 |
Pre-syncope Symptoms |
Lightheadedness |
74 (16.2%) |
|
Nausea |
42 (9.2%) |
||
|
Sweating |
35 (7.6%) |
||
|
Palpitations |
35 (7.6%) |
||
|
Vision abnormalities |
51 (11.1%) |
||
|
Hearing abnormalities |
31 (6.8%) |
||
|
Pallor |
40 (8.8%) |
||
|
Rising sensation from abdomen |
15 (3.3%) |
||
|
4 |
Triggers |
Supine position |
19 (4.2%) |
|
Sitting |
12 (2.6%) |
||
|
Standing for some period |
43 (9.4%) |
||
|
Fear, pain, instrumentation |
24 (5.3%) |
||
|
During physical exercise |
9 (1.9%) |
||
|
After cessation of exercise |
5 (1.1%) |
||
|
During fever |
6 (1.3%) |
||
|
Heat, warmth, hot bath |
21 (4.6%) |
||
|
Sight of blood |
15 (3.3%) |
||
|
5 |
Investigations (n=8) |
CBC |
5 (62.5%) |
|
Glucose level |
5 (62.5%) |
||
|
24-ECG |
1 (12.5%) |
||
|
EEG |
0 (0%) |
||
|
Electrolytes |
2 (25%) |
||
|
Urinalysis |
0 (0%) |
||
|
Echocardiogram |
0 (0%) |
||
|
Tilt table |
0 (0%) |
||
|
EPS |
0 (0%) |
||
|
Exercise test |
0 (0%) |
Table 3: Descriptive table for vasovagal syncope
Note: 448 who are not clinically diagnosed include those who don’t suffer from vvs, one participant may have more than one trigger, pre-episode symptom, and investigation. Investigations are among the 8 who were clinically diagnosed. CBC: complete blood count, 24-ECG: electrocardiogram, EEG: electroencephalogram, EPS: electrophysiological study
|
Variable |
Category (Reference) |
Count (%) |
p-Value (Sig.) |
Odds Ratio (Exp(B)) |
Chi-Square (Wald) |
|
Sleep Hours |
— (continuous) |
— |
0.107 |
0.873 |
2.597 |
|
Caffeine |
Yes (Reference) |
364 (79.8%) |
— |
— |
— |
|
No |
88 (19.3%) |
0.639 |
0.855 |
0.219 |
|
|
Gums |
Yes (Reference) |
184 (40.4%) |
— |
— |
— |
|
No |
267 (58.6%) |
0.548 |
1.175 |
0.36 |
|
|
Stress Rate |
3 (Reference) |
150 (32.9%) |
— |
— |
— |
|
1 |
88 (19.3%) |
0.144 |
8.567 |
2.14 |
|
|
2 |
214 (46.9%) |
0.249 |
5.249 |
1.327 |
|
|
Physical Activity |
Daily (Reference) |
90 (19.7%) |
— |
— |
— |
|
Never |
52 (11.4%) |
0.581 |
0.782 |
0.304 |
|
|
Occasionally |
196 (43.0%) |
0.029 |
0.457 |
4.744 |
|
|
Weekly |
110 (24.1%) |
0.004 |
0.295 |
8.189 |
|
|
Family Hx (VVS) |
No (Reference) |
407 (89.3%) |
— |
— |
— |
|
Yes |
42 (9.2%) |
< 0.001 |
8.658 |
29.921 |
Table 4: Multivariable logistic regression analysis of factors associated with Vasovagal Syncope (VVS)
DISCUSSION
In this cross-sectional analysis involving 456 medical students, interns, and residents from multiple hospitals in Jordan, we examined the hypothesis that smoking might reduce the occurrence of vasovagal syncope (VVS) through its sympathomimetic effects. However, statistical analysis did not reveal a significant association. When vaping was evaluated separately, a borderline trend was observed (p = 0.056). Several variables were identified as significant predictors of VVS, including female sex (p = 0.008), positive family history (p < 0.001), and physical activity levels (weekly exercise: p = 0.004; occasional exercise: p = 0.029).
Among the 121 smokers included in the study, 22 (18.2%) reported VVS symptoms, compared to 72 of 335 non-smokers (21.5%), with no statistically significant difference (p = 0.188). Nevertheless, the near-significant finding related to vaping warrants further investigation in larger populations, as differences in nicotine delivery may play a role. Vaping may induce more rapid sympathetic activation compared to conventional cigarettes [18], and previous studies have highlighted its effects on the autonomic nervous system, including the potential to provoke tachycardia [12].
The higher prevalence of VVS among females compared to males, previously reported in the literature, was also confirmed in our findings (p = 0.008). This may be explained by physiological differences, including relatively higher vagal tone influenced by estrogen, as well as lower circulating blood volume that reduces cardiac preload [3,9]. Additionally, progesterone has been suggested to affect thresholds for sympathetic withdrawal [19].
Interestingly, the relationship between physical activity and VVS demonstrated a U-shaped pattern. Both daily exercise and complete inactivity showed no significant association, whereas moderate activity levels (occasional and weekly exercise) appeared to have a protective effect (p = 0.004 and p = 0.029, respectively). This may be explained by improved orthostatic tolerance associated with moderate exercise, as reported previously [17,20]. Finally, family history emerged as the strongest predictor of VVS in this study, increasing the likelihood of occurrence by approximately 8.7-fold (p < 0.001). This finding aligns with previous research showing that parental history significantly increases the risk of syncope in offspring [10], highlighting the importance of genetic predisposition. Emerging genome-wide evidence also suggests the involvement of multiple interacting loci in the development of VVS [21].
LIMITATIONS
Firstly, our cohort of medical trainees had a mean age of 23.8 years, which may not represent a broader population of smokers. At younger ages, people tend to have a more robust autonomic regulatory response, and this may mask any potential smoking-related effect. Also, only 1.8% of the people with symptoms of VVS were previously diagnosed by a physician, and even though we used standardized syncope parameter scores, like the Calgary Syncope Score, the lack of head-up tilt testing and real-time hemodynamic monitoring may lead to a degree of diagnostic uncertainty and possibly missing some borderline cases. And lastly, due to the cross-sectional design of the study, the temporal relationship between initiation of smoking and the episodes of syncope is unclear.
Clinical Implications and Future Directions
The observed 8.7-fold increase in VVS risk among individuals with a positive family history highlights the need for heightened clinical awareness. Physicians should exercise increased vigilance when evaluating patients presenting with presyncopal symptoms, particularly those with a relevant family history and female patients, who demonstrated significantly higher susceptibility (p = 0.008). Counseling should also include guidance regarding engagement in potentially high-risk activities.
Moderate physical activity may be beneficial for patients with VVS, especially in cases where reduced orthostatic tolerance contributes to symptom onset. Future research should further explore the effects of vaping in larger and more diverse populations, as the borderline findings in this study, together with existing literature, suggest that different nicotine delivery systems may exert varying influences on autonomic function. Additionally, retrospective cohort studies involving VVS patients who smoke could help clarify the temporal relationship between smoking initiation and syncope onset, thereby addressing current limitations related to causality and patient motivation.
CONCLUSION
Approximately one in five medical trainees reported experiencing VVS episodes. Contrary to our initial hypothesis, smoking was not associated with a decreased prevalence of VVS. The strong associations observed with female sex and positive family history are consistent with existing evidence. These findings do not support a meaningful protective sympathomimetic effect of smoking in this relatively young population.
While no significant relationship was identified between smoking and reduced VVS incidence, the borderline association observed with vaping suggests a possible autonomic influence that warrants further investigation. Female sex, family history, and moderate levels of physical activity emerged as key predictors, with family history conferring an approximately 8.7-fold increased risk.
This study has several limitations, including the relatively young study population, reliance on self-reported symptoms for diagnosis, and the cross-sectional design, which limits causal inference. From a clinical perspective, increased attention should be given to individuals with a familial predisposition particularly females and moderate exercise may be encouraged. Future studies should adopt longitudinal designs to better define the temporal relationship between smoking behavior and syncope onset, as well as to compare the physiological effects of different nicotine delivery methods across broader populations.
COMPETING INTERESTS
None
CONTRIBUTORSHIP
None
ACKNOWLEDGMENT
Lara A. Dmour, Rami Riad for their help in data collection
DISCLAIMER
The views expressed in the submitted article are the author's own and not an official opinion of the institution or funder.
FUNDING
No funding
ETHICAL APPROVAL
Ethical approval was obtained from Jordan University Hospital IRB, code 1667/2025/67
PATIENT AND PUBLIC INVOLVEMENT
No closed involvement
DATA SHARING STATEMENT
Available from the population upon request
REFERENCES
- Moya A, Sutton R. Guidelines for the diagnosis and management of syncope (version 2009): The Task Force for the Diagnosis and Management of Syncope of the European Society of Cardiology (ESC). Eur Heart J. 2009;30(21):2631-71. [Crossref] [Google Scholar] [PubMed]
- Mosqueda-Garcia R, Furlan R, MD JT, Fernandez-Violante R. The elusive pathophysiology of neurally mediated syncope. Circulation.2000;102(23):2898-906. [Crossref] [Google Scholar] [PubMed]
- Cottier C. Vasovagal syncope. Therapeutische Umschau. Revue Therapeutique. 1997;54(3):120-6. [Google Scholar] [PubMed]
- Kaufmann H, Berman J, Oribe E, Oliver J. Possible increases in EDRF/NO in neurally mediated syncope. Clin Auton Res.1993;3:77. [Google Scholar]
- Rea RF, Thames MD. Neural control mechanisms and vasovagal syncope. J Cardiovasc Electrophysiol. 1993;4(5):587-95. [Crossref] [Google Scholar] [PubMed]
- Thorén P. Role of cardiac vagal C-fibers in cardiovascular control. Rev Physiol Biochem Pharmacol, 2005;86(21):1-94.[Crossref] [Google Scholar] [PubMed]
- Zyśko D, Szewczuk-Bogusławska M, Kaczmarek M, Agrawal AK, Rudnicki J, et, al. Reflex syncope, anxiety level, and family history of cardiovascular disease in young women: Case–control study. Ep Europace. 2014;17(2):309-13.[Crossref] [Google Scholar] [PubMed]
- Serletis A, Rose S, Sheldon AG, Sheldon RS. Vasovagal syncope in medical students and their first-degree relatives. Eur Heart J. 2006;27(16):1965-70. [Crossref] [Google Scholar] [PubMed]
- Alghamdi MA, Alshahrani FA, Aldihan FA, Alamer NM, Al Dihan FA, et, al. Knowledge and awareness of syncope among the population of Riyadh: A cross-sectional study. Cureus. 2022;14(8). [Crossref] [Google Scholar] [PubMed]
- Alrashed FA, Alsubiheen AM, Alshammari H, Mazi SI, Al-Saud SA, et, al. Stress, anxiety, and depression in pre-clinical medical students: Prevalence and association with sleep disorders. Sustainability. 2022;14(18):11320. [Google Scholar]
- Prijić Ž, Igić R. Cigarette smoking and medical students. J BUON. 2021;26(5):1709-8. [Google Scholar] [PubMed]
- Jamjoom AA, Nikkar-Esfahani A, Fitzgerald JE. Operating theatre related syncope in medical students: a cross sectional study. BMC Medical Education. 2009;9(1):14.[Crossref] [Google Scholar] [PubMed]
- Niedermaier ON, Smith ML, Beightol LA, Zukowska-Grojec Z, Goldstein DS, et, al. Influence of cigarette smoking on human autonomic function. Circulation. 1993;88(2):562-71. [Crossref] [Google Scholar] [PubMed]
- Ragab EA, Dafallah MA, Salih MH, Osman WN, Osman M, et, al. Stress and its correlates among medical students in six medical colleges: An attempt to understand the current situation. Middle East Curr. Psychiatry. 2021;28(1):75. [Google Scholar]
- Abdulghani HM, Irshad M, Al Zunitan MA, Al Sulihem AA, Al Dehaim MA, et, al. Prevalence of stress in junior doctors during their internship training: a cross-sectional study of three Saudi medical colleges’ hospitals. Neuropsychiatr Dis Treat. 2014:1879-86. [Crossref] [Google Scholar] [PubMed]
- Brignole M, Moya A, De Lange FJ, Deharo JC, Elliott PM, et, al. 2018 ESC Guidelines for the diagnosis and management of syncope. Kardiol Pol. 2018;76(8):1119-98. [Crossref] [Google Scholar] [PubMed]
- Sheldon R. Syncope diagnostic scores. Prog Cardiovasc Dis. 2013;55(4):390-5. [Crossref] [Google Scholar] [PubMed]
- Borghi C, Tsioufis K, Agabiti-Rosei E, Burnier M, Cicero AF, et, al. Nutraceuticals and blood pressure control: a European Society of Hypertension position document. J Hypertens. 2020;38(5):799-812. [Crossref] [Google Scholar] [PubMed]
- Rocchi C, Placidi F, Del Bianco C, Liguori C, Pisani A, et, al. Autonomic symptoms, cardiovascular and sudomotor evaluation in de novo type 1 narcolepsy. Clin Auton Res. 2020;30(6):557-62. [Crossref] [Google Scholar] [PubMed]
- Fu Q, Levine BD. Exercise in the postural orthostatic tachycardia syndrome. Auton Neurosci. 2015;188:86-9. [Crossref] [Google Scholar] [PubMed]
- Sheldon RS, Gerull B. Genetic markers of vasovagal syncope. Autonomic Neuroscience. 2021;235:102871. [Crossref] [Google Scholar] [PubMed]
Article Processing Timeline
| 2-5 Days | Initial Quality & Plagiarism Check |
| 25-35 Days |
Peer Review Feedback |
| 45-60 Days | Total article processing time |
Journal Flyer