ABSTRACT
Heart failure remains a leading cause of morbidity, mortality, and healthcare expenditure worldwide, imposing a growing burden on health systems and societies. Its global prevalence continues to rise due to population aging, increasing rates of hypertension, diabetes, and obesity, and improved survival following acute cardiovascular events [1]. Among its clinical phenotypes, Heart Failure with Preserved Ejection Fraction has emerged as the fastest-growing subtype, characterized by impaired ventricular relaxation and filling despite relatively preserved systolic function. This condition is strongly associated with aging, metabolic dysfunction, systemic inflammation, and structural cardiac remodeling, yet effective disease-modifying therapies remain limited [2]. The burden of heart failure is disproportionately high in low- and middle-income countries, where restricted access to evidence-based treatments, limited infrastructure, and workforce shortages exacerbate health inequities [3]. These disparities highlight the need for integrated global strategies that address both clinical and structural determinants of disease.
Beyond traditional risk factors, emerging evidence underscores the contribution of environmental exposures, socioeconomic conditions, and genetic susceptibility to heart failure development and progression. Expanding genomic and DNA-based research offers opportunities to identify molecular pathways, susceptibility variants, and precision-targeted therapeutic strategies that may improve early detection, risk stratification, and individualized treatment. Addressing the global burden of heart failure aligns with international priorities to reduce noncommunicable diseases and advance equitable healthcare delivery, as emphasized by the World Health Organization [4]. By integrating epidemiology, health disparities, and genomic research frontiers, this review provides a framework for coordinated international action to reduce disease burden and advance sustainable cardiovascular health worldwide.
Keywords: Heart Failure; Heart Failure With Preserved Ejection Fraction; Heart Failure With Reduced Ejection Fraction; Epidemiology; Global Burden; Health Disparities; Low- And Middle-Income Countries; Precision Medicine; DNA Research
INTRODUCTION
Heart failure is a chronic and progressive clinical syndrome resulting from structural or functional cardiac abnormalities that impair ventricular filling or reduce effective cardiac output, representing a major global cause of morbidity and mortality [1]. Among its clinical phenotypes, Heart Failure with Preserved Ejection Fraction has emerged as the fastest-growing subtype worldwide, characterized by impaired myocardial relaxation, ventricular stiffness, systemic inflammation, endothelial dysfunction, and metabolic dysregulation despite relatively preserved systolic performance [2]. Unlike heart failure with reduced ejection fraction, for which multiple disease-modifying therapies are available, preserved-ejection-fraction heart failure remains therapeutically limited, reflecting incomplete understanding of its heterogeneous biological mechanisms. This gap presents substantial opportunity for advanced mechanistic research, including genomic, transcriptomic, and DNA-based investigations aimed at identifying susceptibility variants, molecular pathways, and precision-targeted interventions that may redefine risk stratification and treatment strategies.
The global burden of heart failure continues to rise, affecting approximately 64 million individuals worldwide, driven by population aging, urbanization, and increasing prevalence of hypertension, diabetes, obesity, and cardiometabolic disease [5]. Improved survival following acute cardiovascular events has expanded the population living with chronic cardiac dysfunction, increasing long-term healthcare utilization, recurrent hospitalizations, and economic strain [1]. These burdens are disproportionately concentrated in low- and middle-income regions, where limited access to guideline-directed therapy and diagnostic infrastructure exacerbates outcome disparities [3]. Cardiovascular diseases remain the leading cause of death globally, accounting for approximately 17.9 million deaths annually according to the World Health Organization, with heart failure contributing substantially to this mortality burden [4]. Addressing this challenge aligns with the targets of the United Nations Sustainable Development Goal 3, which emphasizes reducing premature mortality from non-communicable diseases and promoting equitable healthcare access [6-10]. This review synthesizes contemporary global evidence on heart failure epidemiology, disparities, and determinants, while highlighting opportunities for translational research, including molecular and genomic strategies, to support precision medicine, inform policy development, and reduce the worldwide burden of disease.
Global Epidemiology of Heart Failure with Preserved Ejection Fraction
Prevalence
Heart failure with preserved ejection fraction (HFpEF) has emerged as the predominant and fastest-growing phenotype within the global heart failure spectrum. Characterized by impaired ventricular relaxation, increased myocardial stiffness, and elevated filling pressures despite preserved systolic function, HFpEF now accounts for approximately half of all heart failure cases in many high-income regions and continues to increase in prevalence worldwide. This growth reflects population aging, rising rates of obesity and diabetes mellitus, persistent hypertension, improved survival following acute cardiovascular events, and increasing recognition of cardiometabolic comorbidity as central drivers of disease pathophysiology. Unlike heart failure with reduced ejection fraction, HFpEF lacks a long-established therapeutic armamentarium, contributing to ongoing clinical complexity and substantial health system burden.
Figure 1: Rising Global Proportion of HFpEF
In high-income countries, HFpEF prevalence increases steeply with age and is particularly common among individuals older than 70 years. Women are disproportionately affected, especially in later life, reflecting interactions among vascular stiffening, hormonal influences, metabolic dysfunction, and systemic inflammation [11-13]. HFpEF is frequently associated with hypertension, obesity, diabetes mellitus, atrial fibrillation, chronic kidney disease, and other conditions that promote endothelial dysfunction and adverse myocardial remodeling. In the United States and Europe, demographic aging and expanding metabolic disease are expected to further increase the absolute number of individuals living with HFpEF over the coming decades.
In low- and middle-income countries, comprehensive epidemiologic data remain limited; however, rising burdens of hypertension, diabetes, obesity, and ischemic heart disease strongly suggest accelerating HFpEF incidence. Under diagnosis is common due to limited access to echocardiography, biomarker testing, and specialized cardiovascular care. As a result, patients frequently present at advanced stages, with greater symptom burden and higher risk of hospitalization. Earlier onset of cardiometabolic risk factors in these settings may also contribute to premature development of HFpEF, amplifying socioeconomic impact during productive years of life.
Globally, the increasing dominance of HFpEF reflects a broader epidemiologic transition in which aging, metabolic dysfunction, systemic inflammation, and vascular remodeling interact to drive diastolic impairment. These trends underscore the urgent need for improved surveillance systems, enhanced diagnostic capacity, region-specific prevention strategies, and equitable access to emerging evidence-based therapies. Addressing HFpEF as a distinct global cardiovascular priority is essential to mitigating its expanding clinical, economic, and societal burden.
Incidence
The incidence of heart failure with preserved ejection fraction (HFpEF) is rising globally and now constitutes a growing proportion of newly diagnosed heart failure cases, particularly in aging populations. In high-income countries, overall heart failure incidence is approximately 0.2–0.5% annually among adults, increasing markedly with age. HFpEF accounts for an expanding share of these incident cases, driven by population aging and the increasing prevalence of hypertension, obesity, diabetes mellitus, and other cardiometabolic conditions that promote myocardial stiffness and diastolic dysfunction. Improved survival after acute cardiovascular events has also increased the number of individuals at risk for developing HFpEF through cumulative structural and functional cardiac remodeling.
In low- and middle-income countries, reliable incidence data remain limited due to underdiagnosis and constrained access to diagnostic tools, including echocardiography and natriuretic peptide testing. Nevertheless, rapid urbanization and rising cardiometabolic risk factors strongly indicate increasing HFpEF incidence in these regions. Patients frequently present at younger ages and more advanced stages of disease, reflecting delayed detection and limited preventive care. These patterns contribute to higher morbidity and premature mortality Strengthening surveillance systems, improving early detection in primary care, and integrating risk-based screening strategies are essential to accurately monitor HFpEF incidence and reduce disparities across regions.
Mortality and Morbidity
Heart failure with preserved ejection fraction (HFpEF) is associated with substantial morbidity and persistently high mortality despite therapeutic advances. Long-term survival remains limited, with approximately half of patients experiencing major clinical deterioration or death within five years of diagnosis. Outcomes are strongly influenced by age, comorbidity burden, socioeconomic factors, and access to timely, evidence-based care. Unlike heart failure with reduced ejection fraction, where pump failure and arrhythmic death are prominent, HFpEF is characterized by recurrent hospitalizations, progressive exercise intolerance, and declining functional capacity. Repeated admissions, often driven by congestion and comorbid conditions, represent the primary contributor to healthcare utilization and cost.
Hospitalization risk in HFpEF is closely linked to hypertension, obesity, diabetes mellitus, atrial fibrillation, chronic kidney disease, and systemic inflammation. Multimorbidity accelerates structural cardiac remodeling, vascular stiffening, and diastolic dysfunction, reinforcing a cycle of decompensation. Beyond clinical outcomes, HFpEF substantially reduces quality of life, limits physical independence, increases caregiver burden, and contributes to long-term disability and productivity loss.
Disparities in morbidity and mortality are most pronounced in low- and middle-income countries, where delayed diagnosis, limited access to diagnostic imaging, restricted availability of evidence-based therapies, and workforce shortages contribute to worse outcomes. Patients frequently present at advanced stages, resulting in higher rates of preventable hospitalization and premature death. Strengthening primary care integration, expanding access to essential therapies, and implementing scalable models such as community-based management, task-sharing, and digital health–supported monitoring are critical to reducing avoidable admissions and improving continuity of care. Aligning these strategies with global non-communicable disease priorities is essential to narrowing outcome disparities and mitigating the global impact of HFpEF
Risk Factors and Determinants
Heart failure with preserved ejection fraction (HFpEF) arises from a multifactorial interplay of non-modifiable biological determinants and population-level influences that collectively shape disease susceptibility, structural remodeling, and clinical progression. Age is the most powerful risk factor, with incidence increasing markedly after 65 years. Aging is associated with progressive myocardial fibrosis, increased ventricular and arterial stiffness, impaired diastolic relaxation, endothelial dysfunction, and heightened systemic inflammation. These changes promote elevated filling pressures despite preserved systolic function, creating the hemodynamic substrate characteristic of HFpEF. In addition, aging is frequently accompanied by multimorbidity, including hypertension, diabetes mellitus, chronic kidney disease, and atrial fibrillation, which further accelerate adverse cardiac remodeling and functional decline.
Figure 2: Major Risk Factors for HFpEF
Sex differences are equally important in defining HFpEF epidemiology and phenotype. Women are disproportionately affected, particularly in older age groups. This pattern reflects interactions among hormonal transitions, vascular stiffening, metabolic dysregulation, and distinct myocardial remodeling responses. Compared with men, women with HFpEF more commonly exhibit concentric left ventricular remodeling, increased extracellular matrix deposition, and heightened sensitivity to volume overload. Obesity, insulin resistance, and systemic inflammation are also strongly associated with HFpEF in women, contributing to endothelial impairment and microvascular dysfunction. In contrast, men more frequently develop systolic dysfunction, often linked to ischemic injury and ventricular dilatation.
These age- and sex-related biological differences influence symptom burden, hospitalization risk, exercise intolerance, and long-term outcomes. Recognizing their mechanistic underpinnings is essential for accurate risk stratification, early identification of high-risk individuals, and the development of targeted prevention and therapeutic strategies tailored specifically to HFpEF.
Non-Modifiable Risk Factors
Age: Age is the strongest determinant of heart failure with preserved ejection fraction (HFpEF), with prevalence and incidence increasing markedly after 65 years. Aging is associated with progressive myocardial fibrosis, increased ventricular and arterial stiffness, impaired diastolic relaxation, endothelial dysfunction, and chronic low-grade systemic inflammation. These structural and functional alterations promote elevated filling pressures despite preserved systolic function, creating the hemodynamic foundation for HFpEF. The accumulation of age-related comorbidities further amplifies risk and accelerates clinical progression.
Sex: Sex differences play a central role in HFpEF epidemiology and phenotype. Women are disproportionately affected, particularly in older age groups. This pattern reflects sex-specific differences in cardiac remodeling, vascular biology, and metabolic regulation. Women more commonly exhibit concentric ventricular remodeling, increased extracellular matrix deposition, and heightened susceptibility to hypertension- and obesity-related cardiac changes. In contrast, men more frequently develop systolic dysfunction. These biological distinctions influence symptom presentation, hospitalization risk, and long-term outcomes.
Genetics: Genetic predisposition contributes to HFpEF through both rare and common mechanisms. While monogenic cardiomyopathies are less typical in classic HFpEF, inherited variants influencing myocardial structure, fibrosis pathways, inflammatory signaling, and metabolic regulation may increase susceptibility. Polygenic risk interacts with environmental and cardiometabolic exposures, modulating disease expression, severity, and progression. Understanding genetic contributions is increasingly important for risk stratification and the development of precision medicine strategies in HFpEF.
Modifiable Risk Factors
Hypertension: Hypertension is the most significant modifiable risk factor for heart failure with preserved ejection fraction (HFpEF) globally. Chronic pressure overload promotes concentric left ventricular remodeling, myocardial hypertrophy, interstitial fibrosis, and progressive diastolic dysfunction. In regions with limited treatment coverage and poor blood pressure control, the sustained hemodynamic burden accelerates structural remodeling and substantially increases HFpEF incidence. Effective blood pressure management remains a cornerstone of prevention.
Diabetes Mellitus: Diabetes substantially increases the risk of heart failure, including HFpEF, through metabolic, inflammatory, and microvascular mechanisms. Insulin resistance, myocardial lipid accumulation, oxidative stress, and endothelial dysfunction contribute to impaired ventricular relaxation and increased myocardial stiffness. Diabetes also accelerates vascular aging and promotes systemic inflammation, both of which intensify diastolic impairment and clinical progression.
Obesity and Metabolic Syndrome: Obesity is a central driver of the modern HFpEF epidemic. Excess adiposity contributes to systemic inflammation, expanded plasma volume, increased cardiac workload, and adverse ventricular remodeling. Visceral fat is particularly associated with insulin resistance, cytokine activation, and endothelial dysfunction, all of which promote diastolic dysfunction. Metabolic syndrome further compounds risk by clustering hypertension, dyslipidemia, and glucose intolerance, creating a high-risk cardiometabolic phenotype strongly linked to HFpEF.
Coronary Artery Disease: While more strongly associated with heart failure with reduced ejection fraction, coronary artery disease remains an important contributor to HFpEF, particularly in aging populations. Ischemic injury, even when subclinical, can impair myocardial relaxation and exacerbate diastolic dysfunction. Coexisting ischemia also worsens symptoms and increases hospitalization risk in patients with established HFpEF.
Social Determinants of Health
Social determinants play a critical role in shaping the risk, diagnosis, treatment, and outcomes of heart failure with preserved ejection fraction (HFpEF). Poverty, limited access to primary and specialized care, low health literacy, and geographic barriers contribute to delayed recognition of symptoms, inadequate control of hypertension and diabetes, and suboptimal implementation of preventive strategies. These factors disproportionately affect populations in low- and middle-income countries, where diagnostic infrastructure and long-term disease management resources are often limited. Urban–rural disparities further exacerbate inequities by restricting access to echocardiography, laboratory testing, and guideline-directed therapies.
Collectively, epidemiologic trends demonstrate that HFpEF is not solely a biological condition but also a socially patterned disease influenced by structural inequities. The interaction between non-modifiable determinants (age, sex, genetic susceptibility), modifiable cardiometabolic risks, and health system capacity determines patterns of incidence, clinical phenotype, hospitalization risk, and survival. In resource-constrained settings, delayed diagnosis and limited access to evidence-based interventions result in more advanced disease at presentation and higher preventable morbidity and mortality.
Recognizing HFpEF as a life-course condition shaped by structural, clinical, and policy environments underscores the need for integrated strategies that extend beyond individual-level treatment. Strengthening surveillance systems, enhancing early risk stratification in primary care, and embedding HFpEF prevention within broader non-communicable disease frameworks are essential to reducing disparities. Aligning these efforts with global health priorities set forth by international health governance bodies reinforces the importance of translating epidemiologic evidence into equitable, scalable cardiovascular health policies.
Figure 3: Age-Related Increase in Filling Pressures
Global Burden and Healthcare Impact
Heart failure (HF) imposes one of the greatest economic and societal burdens among cardiovascular diseases, reflecting its chronic course, high rates of hospitalization, and progressive functional decline. Direct medical costs are substantial and continue to rise, driven by recurrent inpatient admissions, advanced pharmacologic and device therapies, and long-term outpatient management. In the United States alone, annual HF-related expenditures exceed $30 billion, underscoring the strain placed on even well-resourced health systems [14]. Beyond direct healthcare spending, HF generates profound indirect costs through lost productivity, disability, caregiver burden, and diminished quality of life, effects that extend across households and communities. As populations age and survival from acute cardiovascular events improves, the cumulative economic impact of HF is projected to increase further, positioning it as a critical threat to health system sustainability worldwide.
- Direct medical costs are substantial; in the US, HF costs exceed $30 billion annually [15].
- Indirect costs include lost productivity, caregiver burden, and reduced quality of life.
- LMICs face disproportionate challenges due to limited access to medications, devices, and specialized care.
The global impact of HF is most severe in low- and middle-income countries (LMICs), where constrained health system capacity magnifies both clinical and economic consequences. Limited access to essential medications, advanced diagnostics, device therapies, and specialized cardiovascular care contributes to delayed diagnosis, higher hospitalization rates, and excess premature mortality. These challenges are compounded by inadequate insurance coverage, workforce shortages, and weak social support systems, shifting much of the financial and caregiving burden to patients and families. Addressing the global HF burden therefore requires more than expanding clinical services; it demands coordinated investment in health system strengthening, equitable access to essential therapies, and integration of HF care within broader non-communicable disease frameworks. Scalable approaches, including community-based management, task-shifting, and digital health–enabled care models offer pathways to mitigate costs while improving outcomes. Framing HF as both a clinical and socioeconomic challenge reinforces the imperative for policy-driven solutions that protect health system resilience and promote cardiovascular health equity across diverse global contexts [16-20].
Implications for WHO and UN Health Policy
Heart failure (HF) represents a critical but often underrecognized component of the global non-communicable disease (NCD) agenda, demanding coordinated policy responses at national and international levels. As the burden of HF continues to rise across diverse regions, its effective prevention and management require alignment with global health governance structures led by the World Health Organization and the United Nations. Strengthening surveillance systems, improving prevention and risk factor control, and ensuring equitable access to evidence-based therapies are central to mitigating HF-related morbidity, mortality, and economic impact. Integrating HF explicitly within broader NCD strategies offers an opportunity to leverage existing frameworks, enhance data comparability, and drive scalable interventions that address both clinical needs and structural health inequities, particularly in low- and middle-income countries (LMICs).
Surveillance and Data Collection
- Establishing standardized HF registries is critical, particularly in LMICs, to monitor incidence, prevalence, outcomes, and therapy utilization.
- Integration with WHO NCD monitoring frameworks can enhance global data quality [21-24].
Prevention and Risk Factor Control
- Population-level interventions targeting hypertension, diabetes, obesity, and smoking are essential.
- Early detection and management of myocardial infarction, valvular disease, and cardiomyopathies can reduce HF incidence.
Access to Guideline-Directed Medical Therapy (GDMT)
- WHO and UN initiatives should prioritize equitable access to ARNI, SGLT2 inhibitors, beta-blockers, and mineralocorticoid receptor antagonists, particularly in LMICs [25-28].
- Telemedicine, task-shifting, and community-based programs can expand access.
Integration with Sustainable Development Goals (SDGs)
- HF management aligns with SDG 3 and the broader NCD Global Action Plan, contributing to reduced premature mortality and improved health equity.
Effective policy action on HF must prioritize robust surveillance and data collection through standardized registries, especially in LMICs where epidemiologic data remain sparse and fragmented. Integration of HF metrics into existing WHO NCD monitoring frameworks can improve global data quality, inform resource allocation, and guide evidence-based decision-making. Prevention strategies focused on population-level control of hypertension, diabetes, obesity, and tobacco use alongside early detection and treatment of myocardial infarction, valvular disease, and cardiomyopathies are essential to curbing future HF incidence. Equally critical is ensuring equitable access to guideline-directed medical therapy, including angiotensin receptor–neprilysin inhibitors, sodium–glucose cotransporter 2 inhibitors, beta-blockers, and mineralocorticoid receptor antagonists, supported by innovative delivery models such as telemedicine, task-shifting, and community-based care. Framing HF management within the context of Sustainable Development Goal 3 and the global NCD action plan reinforces its relevance to reducing premature mortality, strengthening health systems, and advancing health equity. Embedding HF within international policy priorities thus represents a pragmatic and necessary step toward achieving sustainable cardiovascular health gains worldwide.
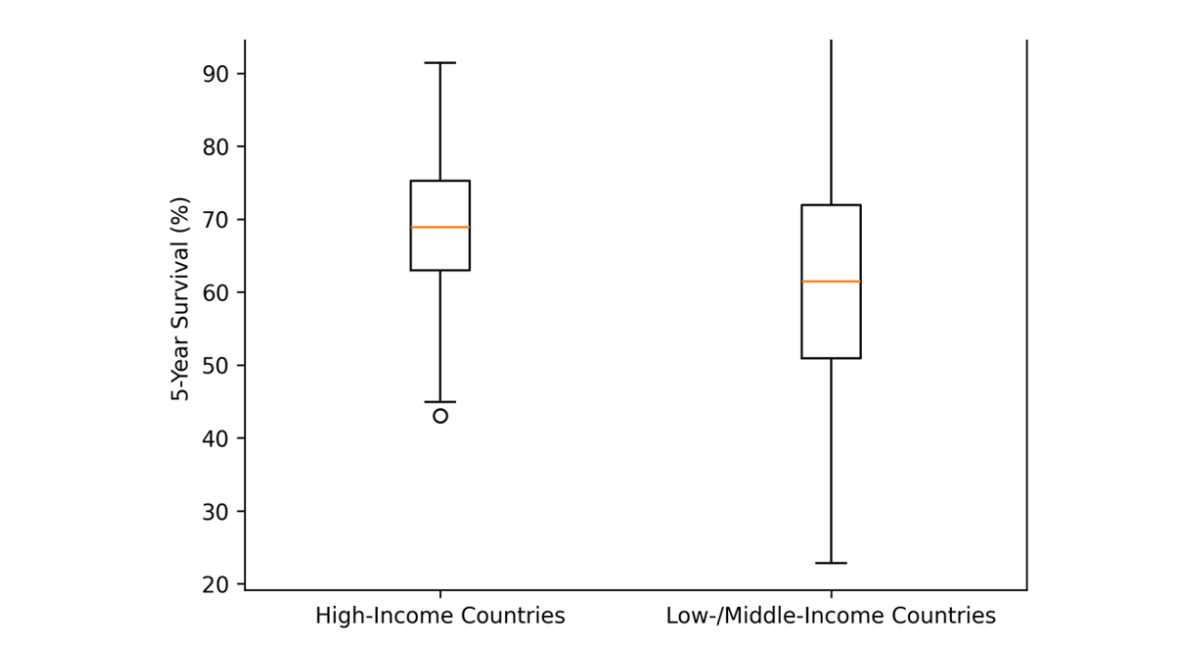
Figure 4: Outcome Disparities in HFpEF
Research Gaps and Future Directions in Heart Failure with Preserved Ejection Fraction: Strengthening Global Surveillance through Genomic and DNA-Based Integration
Despite substantial advances in diagnosis and therapy, major gaps remain in the global understanding of heart failure with preserved ejection fraction (HFpEF), particularly in epidemiology, mechanistic biology, risk stratification, and intervention effectiveness across diverse populations. Low- and middle-income countries remain underrepresented in population-level studies, limiting accurate estimates of incidence, prevalence, phenotype distribution, and outcomes. This data gap constrains evidence-based resource allocation and weakens global surveillance frameworks. In addition, clinical trials frequently exclude women, older adults, and socioeconomically disadvantaged populations groups that bear the highest burden of HFpEF thereby limiting external validity and equitable translation of guideline-directed therapies.
Future research must integrate genomic and DNA-based approaches to advance precision risk prediction and mechanistic understanding. HFpEF is increasingly recognized as a genetically heterogeneous syndrome in which polygenic susceptibility interacts with cardiometabolic exposures, systemic inflammation, and environmental determinants. Large-scale genomic studies, multi-omics integration, and DNA-based risk profiling may enable earlier identification of high-risk individuals, refine phenotypic subtyping, and support development of targeted therapeutics. Incorporating ancestry-diverse genomic datasets is essential to prevent widening disparities in precision medicine.
In parallel, culturally tailored and cost-effective interventions are needed to ensure scalability across health systems with varying resources. Community-based prevention strategies, integration of HFpEF management into primary care, and strengthening of surveillance systems in low-resource settings are critical priorities. Emerging technologies, including digital health platforms, biomarker-guided diagnostics, and artificial intelligence enabled risk modeling offer transformative potential for early detection, personalized management, and population-level monitoring [29-32]. Aligning these research priorities with global non-communicable disease frameworks and international health governance strategies will generate actionable evidence, reduce inequities, and accelerate sustainable improvements in HFpEF outcomes worldwide.
Highlights
This review uniquely centers heart failure with preserved ejection fraction (HFpEF) as the dominant and fastest-growing global heart failure phenotype, integrating epidemiology, risk architecture, health systems, and policy within a single unified framework. Rather than summarizing existing literature, it synthesizes global prevalence and incidence trends, sex-specific patterns, cardiometabolic determinants, and structural health disparities into one coherent model that connects biological mechanisms with population-level outcomes. The review emphasizes HFpEF as a life-course, multisystem disorder driven by aging, metabolic dysfunction, vascular stiffness, systemic inflammation, and genetic susceptibility, thereby moving beyond traditional pump-centered paradigms [33-35].
A distinguishing feature of this manuscript is its integration of genomic and DNA-based perspectives into global HFpEF strategy, highlighting opportunities for precision risk stratification, ancestry-diverse genomic research, and future targeted therapies. In addition, the review aligns epidemiologic evidence with actionable policy recommendations, linking prevention strategies, guideline-directed therapies, digital health innovation, and health system strengthening to international non-communicable disease frameworks. By bridging molecular mechanisms, clinical management, population surveillance, and World Health Organization and United Nations priorities, this work provides a multidimensional perspective that is both scientifically rigorous and policy-relevant. This integrated approach, combining epidemiology, disparities, genomics, precision medicine, and global health strategy, distinguishes the review from conventional narrative summaries and offers readers a comprehensive, forward-looking framework for understanding and addressing HFpEF worldwide.
CONCLUSION
Heart failure (HF), particularly heart failure with preserved ejection fraction (HFpEF), represents an escalating global public health challenge with profound clinical, economic, and societal consequences. Its increasing prevalence reflects demographic aging, population-level exposure to hypertension, diabetes mellitus, and obesity, and improved survival following acute cardiovascular events. These epidemiologic shifts have accelerated the transition toward HFpEF as the dominant phenotype in older adults, especially women, characterized by systemic inflammation, metabolic dysregulation, endothelial dysfunction, and vascular stiffness. In contrast, heart failure with reduced ejection fraction more commonly follows ischemic injury, underscoring the mechanistic heterogeneity of the syndrome. Persistent disparities between high-income and low- and middle-income countries highlight structural inequities in preventive care, diagnostic infrastructure, and access to evidence-based therapies, contributing to differences in disease onset, severity, and outcomes.
Reducing the global burden of HF requires integrated strategies that combine surveillance, prevention, clinical management, and policy reform. Standardized population-based registries and alignment with international non-communicable disease monitoring frameworks are essential for accurate epidemiologic assessment and equitable resource allocation. Population-level interventions targeting modifiable cardiometabolic risk factors, particularly blood pressure control, glycemic management, obesity prevention, and tobacco reduction, are central to limiting HF incidence and progression. Expanding equitable access to guideline-directed therapies, supported by primary care integration, telemedicine, community-based models, and task-sharing approaches, is critical in resource-constrained.
ABBREVIATIONS
To ensure clarity, consistency, and readability throughout the manuscript, the following abbreviations are used. Providing a consolidated list facilitates comprehension for readers across disciplines and supports standardized terminology in accordance with international publishing conventions.
HF – Heart Failure: A clinical syndrome characterized by structural and/or functional impairment of cardiac function, leading to inadequate systemic perfusion or elevated intracardiac pressure.
HFpEF – Heart Failure with Preserved Ejection Fraction: A subtype of heart failure in which left ventricular ejection fraction is preserved, but diastolic dysfunction, impaired relaxation, and elevated filling pressures result in clinical symptoms and signs of heart failure.
HFrEF – Heart Failure with Reduced Ejection Fraction: A subtype of heart failure characterized by reduced left ventricular ejection fraction, typically associated with impaired systolic function and ventricular remodeling.
LMICs – Low- and Middle-Income Countries: Nations classified by the World Bank according to gross national income per capita, often characterized by limited healthcare resources and higher barriers to diagnostic and therapeutic access.
HICs – High-Income Countries: Nations classified by the World Bank with high gross national income per capita and generally greater access to advanced healthcare infrastructure and therapies.
WHO – World Health Organization: A specialized agency of the United Nations responsible for international public health coordination, standards, and policy guidance.
UN – United Nations: An intergovernmental organization that promotes international cooperation, sustainable development, and global policy initiatives, including health-related goals.
GDMT – Guideline-Directed Medical Therapy: Evidence-based pharmacologic and therapeutic interventions recommended in clinical guidelines for the management of heart failure.
SDGs – Sustainable Development Goals: A set of 17 global objectives established by the United Nations to promote health, equity, economic stability, and sustainable development, including targets relevant to non-communicable disease reduction.
All abbreviations are defined at first mention within the text and are included in this section for reference to promote transparency and ease of interpretation. This standardized terminology ensures consistency throughout the manuscript and aligns with established guidelines for scientific reporting.
REFERENCES
- Savarese G, Lund LH. Global public health burden of heart failure. Cardiac failure review. 2017r;3(1):7. [Crossref] [Google Scholar] [PubMed]
- Dunlay SM, Roger VL, Redfield MM. Epidemiology of heart failure with preserved ejection fraction. Nature Reviews Cardiology. 2017;14(10):591-602. [Crossref] [Google Scholar] [PubMed]
- Yayla Ç, Arslan U, editors. Kalp Yetersizliğinde Pratik Bilgiler. Akademisyen Kitabevi; 2021. [Crossref] [Google Scholar]
- World Health Organization. cardiovascular diseases (CVDs) fact sheet. World Health Organ. 2020;42(1):207-16. [Google Scholar]
- Roth GA, Mensah GA, Johnson CO, Addolorato G, Ammirati E, Baddour LM, et al. Global burden of cardiovascular diseases and risk factors, 1990–2019: update from the GBD 2019 study. Journal of the American college of cardiology. 2020;76(25):2982-3021. [Crossref] [Google Scholar]
- Emmons-Bell S, Johnson C, Roth G. Prevalence, incidence and survival of heart failure: a systematic review. Heart. 2022;108(17):1351-60. [Crossref] [Google Scholar] [PubMed]
- Savarese G, Becher PM, Lund LH, Seferovic P, Rosano GM, Coats AJ. Global burden of heart failure: a comprehensive and updated review of epidemiology. Cardiovascular research. 2022 ;118(17):3272-87. [Crossref] [Google Scholar] [PubMed]
- Chen QF, Chen L, Katsouras CS, Liu C, Shi J, Liang D, Xiang G, Zhu H, Liao H, Lin W, Zhou X. Global burden of heart failure and its underlying causes in 204 countries and territories, 1990–2021. European Heart Journal-Quality of Care and Clinical Outcomes. 2025;11(4):493-509. [Crossref] [Google Scholar] [PubMed]
- Groenewegen A, Rutten FH, Mosterd A, Hoes AW. Epidemiology of heart failure. European journal of heart failure. 2020;22(8):1342-56. [Crossref] [Google Scholar] [PubMed]
- United Nations. Transforming our world: the 2030 Agenda for Sustainable Development. UN; 2015. [Crossref] [Google Scholar]
- Lam CS, Arnott C, Beale AL, Chandramouli C, Hilfiker-Kleiner D, Kaye DM,et al. Sex differences in heart failure. European heart journal. 2019;40(47):3859-68c. [Crossref] [Google Scholar] [PubMed]
- Vogel B, Acevedo M, Appelman Y, Merz CN, Chieffo A, Figtree GA, et al. The Lancet women and cardiovascular disease Commission: reducing the global burden by 2030. The Lancet. 2021;397(10292):2385-438. [Crossref] [Google Scholar]
- Regitz-Zagrosek V. Sex differences in heart failure. Int J Heart Fail. 2020;2:157–181. [PubMed]
- Cook C, Cole G, Asaria P, Jabbour R, Francis DP. The annual global economic burden of heart failure. International journal of cardiology. 2014;171(3):368-76. [Crossref] [Google Scholar] [PubMed]
- Organización Mundial de la Salud. Global Status Report on Noncommunicable Diseases. World Health Organization; 2011. [Google Scholar]
- McDonagh TA, Metra M, Adamo M, Gardner RS, Baumbach A, Böhm M. ESC guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur Heart J2021; 42: 3599–3726. 2021. [Crossref] [Google Scholar] [PubMed]
- Heidenreich PA, Bozkurt B, Aguilar D, Allen LA, Byun JJ, Colvin MM, et al . 2022 AHA/ACC/HFSA guideline for the management of heart failure: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Journal of the American College of Cardiology. 2022 May 3;79(17):e263-421. [Crossref] [Google Scholar] [PubMed]
- Virani SS, et al. 2023 AHA/ACC guideline for primary prevention. Circulation. 2023. [Google Scholar] [PubMed]
- Bertram Pitt MD, Zannad F, Remme WJ, Cody R, CASTAIGNE A, Perez A, et al. The effect of spironolactone on morbidity and mortality in patients with severe heart failure. N Engl J Med. 1999;341(10):709-17. [Crossref] [Google Scholar] [PubMed]
- McMurray JJ, Packer M, Desai AS, Gong J, LeAowitz MP, Rizkala AR, et al. PARADIGM-HF. N Engl J Med. 2014;371(11):993-1004. [Crossref] [Google Scholar] [PubMed]
- McMurray JJ, Solomon SD, Inzucchi SE, Køber L, Kosiborod MN, Martinez FA, et al. Dapagliflozin in patients with heart failure and reduced ejection fraction. New England Journal of Medicine. 2019;381(21):1995-2008. [Crossref] [Google Scholar]
- Packer M, Anker S, Butler J. EMPEROR-reduced. N Engl J Med. 2020;383:1413-24. [Google Scholar]
- Anker SD, Butler J, Filippatos G, Ferreira JP, Bocchi E, Böhm M, et al. Empagliflozin in heart failure with a preserved ejection fraction. New England Journal of Medicine. 2021;385(16):1451-61. [Google Scholar]
- Solomon SD, McMurray JJ, Claggett B, de Boer RA, DeMets D, Hernandez AF, et al. Dapagliflozin in heart failure with mildly reduced or preserved ejection fraction. New England Journal of Medicine. 2022;387(12):1089-98. [Google Scholar]
- Zannad F, Ferreira JP, Pocock SJ, Anker SD, Butler J, Filippatos G, et al. SGLT2 inhibitors in patients with heart failure with reduced ejection fraction: a meta-analysis of the EMPEROR-Reduced and DAPA-HF trials. The Lancet.;396(10254):819-29. [Google Scholar]
- Ali AE, Mazroua MS, Elsaban M, Najam N, Kothari AS, Mansoor T, et al. Effect of dapagliflozin in patients with heart failure: a systematic review and meta-analysis. Global heart. 2023;18(1):45. [Crossref] [Google Scholar] [PubMed]
- Alrasheed RR, Altaf AF, Althurwi AH, Alrodan SF, Asiri MH, Alsaluli BA, et al. Heart Failure Outcomes with SGLT2 Inhibitors in Adults with Type 2 Diabetes: A Systematic Review and Meta-Analysis. Medicina. 2025;62(1):69. [Crossref] [Google Scholar] [PubMed]
- Metra M, Tomasoni D, Adamo M, Amir O, Anker SD, Bayes-Genis A, et al. SGLT2 inhibitors for the prevention and treatment of heart failure: A scientific statement of the HFA and the HFAI. ESC Heart Failure. 2025;12(6):3806-25. [Crossref] [Google Scholar] [PubMed]
- Lüscher TF, Wenzl FA, D’Ascenzo F, Friedman PA, Antoniades C. Artificial intelligence in cardiovascular medicine: clinical applications. European heart journal. 2024;45(40):4291-304. [Crossref] [Google Scholar] [PubMed]
- Deo RC. Machine learning in medicine. Circulation. 2015;132(20):1920-30. [Crossref] [Google Scholar]
- Topol EJ. High-performance medicine: the convergence of human and artificial intelligence. Nature medicine. 2019;25(1):44-56. [Crossref] [Google Scholar] [PubMed]
- Asta L, Pisano C, Sbrigata A, Raffa GM, Scola L, Balistreri CR. Biomarkers in heart failure: a review and a wish. International Journal of Molecular Sciences. 2025;26(16):8046. [Crossref] [Google Scholar] [PubMed]
- Hershberger RE, Givertz MM, Ho CY, Judge DP, Kantor PF, McBride KL, Morales A, Taylor MR, Vatta M, Ware SM. Genetic evaluation of cardiomyopathy—a Heart Failure Society of America practice guideline. Journal of cardiac failure. 2018;24(5):281-302. [Crossref] [Google Scholar]
- Jiang Y, Schmidt RH, Zhao Y, Reif JC. A quantitative genetic framework highlights the role of epistatic effects for grain-yield heterosis in bread wheat. Nature genetics. 2017;49(12):1741-6. [Crossref] [Google Scholar] [PubMed]
- Roth GA, Dorsey H, Decleene N, Razo C, Stark B, Johnson C. The global burden of heart failure: a systematic analysis for the Global Burden of Disease Study 2021. European Heart Journal. 2023;44-876. [Crossref] [Google Scholar]
Article Processing Timeline
| 2-5 Days | Initial Quality & Plagiarism Check |
| 25-35 Days |
Peer Review Feedback |
| 45-60 Days | Total article processing time |
Journal Flyer