ABSTRACT
Background
Heart failure (HF) remains among the most consequential syndromes in cardiovascular medicine, affecting an estimated 64 million individuals worldwide and accounting, in developed health systems, for more hospital admissions than any other single diagnosis. The 2021 European Society of Cardiology (ESC) guidelines formalised the tripartite classification of HF by left ventricular ejection fraction (LVEF): HF with reduced EF (HFrEF, LVEF < 40%), HF with mildly reduced EF (HFmrEF, LVEF 40-49%), and HF with preserved EF (HFpEF, LVEF ≥ 50%). This classification has brought conceptual clarity while also revealing that phenotypic categories are not immutable, and that a patient's position within the EF spectrum may shift substantially over months to years depending on the adequacy of therapy, the underlying aetiology, and the systemic comorbidity burden.
Objectives
This narrative review synthesises current evidence on the natural history, pathophysiological mechanisms, prognostic biomarkers, phenotypic transition dynamics, and pharmacological management of HFrEF, HFmrEF, and HFpEF, with particular emphasis on the cross-phenotype therapeutic activity of sodium-glucose cotransporter-2 (SGLT2) inhibitors.
Data Sources
A structured search of PubMed, MEDLINE, and the Cochrane Central Register of Controlled Trials was conducted for the period 2015-2024 using standardised MeSH terms. Landmark randomised controlled trials (RCTs), pre-specified sub-group analyses, registry-based cohort studies, and consensus guidelines from the ESC, AHA/ACC, and HFA were included.
Results
HFrEF carries the highest short-term mortality risk, with sudden cardiac death accounting for 42% of total deaths; however, reverse cardiac remodelling is achievable in 35-40% of patients under full optimal medical therapy (OMT). HFmrEF demonstrates marked trajectory instability: at two-year follow-up, 35-40% of patients transition toward HFpEF and 25-35% deteriorate toward HFrEF without prompt pharmacological optimisation. HFpEF, now representing up to 52% of all incident HF cases, is driven by systemic microvascular inflammation; its mortality profile now approaches that of HFrEF when adjusted for age and multimorbidity. Across all three phenotypes, SGLT2 inhibitors yield consistent reductions in the composite of cardiovascular death and HF hospitalisation (pooled HR 0.77; 95% CI 0.72-0.83; n = 20,725).
Conclusions
LVEF-based phenotyping provides an indispensable but inherently dynamic framework for clinical decision-making in HF. Therapeutic strategies should be informed by serial echocardiographic reassessment and biomarker monitoring. SGLT2 inhibitors represent the most significant therapeutic advance in HF pharmacotherapy since neurohumoral blockade and warrant early, universal implementation in the eligible HF population.
Keywords: Heart Failure; Ejection Fraction; Hfref; Hfmref; Hfpef; SGLT2 Inhibitors; Cardiac Remodelling; Phenotypic Transition; Biomarkers; Prognosis; Reverse Remodelling
INTRODUCTION
The clinical conceptualisation of heart failure has been transformed over the past quarter-century. What was once treated as a fairly coherent entity defined by dyspnoea, oedema, and reduced cardiac output has been progressively disaggregated into a family of distinct syndromes that share a name and symptom cluster but diverge sharply in their pathophysiology, natural history, and response to therapeutic intervention. The tripartite classification anchored by left ventricular ejection fraction, consolidated in the 2021 ESC guidelines, is the clearest expression of this intellectual evolution [1].
From a global public health standpoint, the numbers are sobering. Somewhere between 64 and 70 million people are living with heart failure at any given moment, and the lifetime risk for an individual reaching the age of 40 in a high-income country now sits at approximately one in five [2]. Five-year mortality, in the range of 50-60% for HFrEF and only marginally lower for HFpEF, compares unfavourably with many common solid tumours. Hospital readmission rates within 30 days of an index HF admission hover around 20-25% across all phenotypes and have proven stubbornly resistant to quality improvement initiatives [2,3].
The EMPEROR-P and DELIVER trials established, as recently as 2021-2022, that SGLT2 inhibitors reduce hard outcomes in HFpEF, completing a sequence of trials that now supports this drug class across the full EF continuum. The implications challenge clinicians to think differently about what HF phenotypes actually represent and how stable those representations are over time [4,5].
The question of temporal stability is among the most clinically consequential and underappreciated aspects of modern HF management. The assumption that a patient classified as HFmrEF at a single echocardiographic encounter will remain HFmrEF is not supported by the longitudinal data [6,7]. Transitions between phenotypic categories are common, bidirectional, and outcome-relevant.
This review addresses the evolutionary trajectories of HF across the EF spectrum with an integrative perspective, treating each phenotype not as a static condition but as a dynamic state with a characteristic natural history that can be modified by therapeutic and biological forces. We examine the mechanisms underlying phenotypic transitions, the biomarker tools available to track them, the current evidence base for pharmacological intervention, and the emerging role of SGLT2 inhibitors as the first true cross-phenotype disease-modifying therapy in HF.
METHODS
A comprehensive narrative review was conducted following a structured search of three major databases PubMed, MEDLINE, and the Cochrane Central Register of Controlled Trials in January 2025. Search terms combined the following MeSH headings and keywords: "Heart Failure", "Ejection Fraction", "Hfref", "Hfmref", "Hfpef", "Preserved Ejection Fraction", "Reduced Ejection Fraction", "Reverse Cardiac Remodelling", "Phenotypic Transition", "SGLT2 Inhibitors", "Dapagliflozin", "Empagliflozin", "Biomarkers", "NT-Probnp" and "Mortality". Boolean operators were applied to refine and cross-reference search combinations.
Selection Process: Studies were eligible for inclusion if they met the following criteria:
(i) randomised controlled trials with enrolment ≥ 500 participants and minimum follow-up of 12 months;
(ii) prospective registry or cohort studies reporting LVEF-stratified clinical outcomes;
(iii) pre-specified sub-group analyses from major trials;
(iv) meta-analyses or systematic reviews published since 2015;
(v) clinical practice guidelines or consensus documents from the ESC, AHA/ACC, or HFA.
Studies limited to pediatric populations, congenital heart disease, or perioperative cardiac failure were excluded. All identified records were screened by title/abstract, followed by full-text review of eligible studies. A total of 20 primary references were selected for inclusion.
Potential Limitations and Selection Bias: As an author-conducted narrative review, this synthesis is subject to potential selection bias. Studies with positive or statistically significant results may have been more readily identified through database searching. Findings from landmark trials conducted predominantly in high-income, European, and North American populations may not be directly generalisable to resource-limited settings. These limitations are acknowledged throughout the text. No ethics approval was required for this narrative review of published data.
RESULTS
The ESC 2021 Tripartite Classification: Rationale, Criteria, and Limitations
The definitive recognition of HF with LVEF 40-49% as a distinct clinical entity came with the 2016 ESC guidelines and was reinforced in 2021 [1]. Before that, clinicians operated with a binary framework that assigned everything above 40% to a preserved-EF category that was never homogeneous. The introduction of HFmrEF was partly an act of intellectual honesty about the heterogeneity lurking in the 40-50% range, and partly a pragmatic response to emerging data suggesting a biological profile more nuanced than either extreme.
The diagnostic criteria for each category reflect this evolution. HFrEF requires only an LVEF below 40% with symptoms and/or signs. HFmrEF and HFpEF require at least one additional structural or functional criterion typically diastolic dysfunction, elevated filling pressures, or structural remodelling along with elevated natriuretic peptides. The HFA-PEFF algorithm provides a five-step diagnostic scoring framework that operationalises these criteria [8-11].
One must acknowledge the limitations of this classification. LVEF is a load-dependent measure, susceptible to variation with changes in preload, afterload, and heart rate. This phenomenon of "reclassification by optimisation" has been documented in registry studies [6,7] and likely contributes to the apparent excess of HFmrEF in cross-sectional surveys. The clinical implication is clear: single-point LVEF measurement is insufficient for reliable phenotypic classification, and serial reassessment under stable, optimally treated conditions is diagnostically imperative.
|
Parameter |
HFrEF |
HFmrEF |
HFpEF |
|
LVEF threshold |
< 40% |
40-49% |
≥ 50% |
|
Symptoms/signs |
Required |
Required |
Required |
|
Structural criteria |
Not required |
≥ 1 abnormality |
≥ 1 abnormality + raised NP |
|
Typical demographic |
Male, < 65 yrs, ischaemic |
Mixed; intermediate |
Female, > 70 yrs, metabolic |
|
Global prevalence |
~50% (stable) |
~15% (stable) |
~35% (rising → ~52%) |
|
5-year all-cause mortality |
50-60% |
40-50% |
40-50% |
|
Predominant death mode |
Sudden cardiac (42%) |
Mixed SCD/pump |
Non-cardiovascular (46%) |
|
30-day rehospitalisation |
~24% |
~21% |
~20% |
|
NT-proBNP (median) |
> 2,500 pg/mL |
~1,450 pg/mL |
800-1,200 pg/mL |
|
Comorbidity burden |
Moderate |
Moderate-high |
High (metabolic) |
Table 1: ESC 2021 Diagnostic Criteria and Comparative Clinical Profile by HF Phenotype
NP = Natriuretic Peptide; SCD = Sudden Cardiac Death; LVEF = Left Ventricular Ejection Fraction. Adapted from: McDonagh TA ESC Guidelines [1].
Clinical Application Summary: The ESC tripartite framework informs both diagnosis and prescribing. For HFrEF, it triggers evidence-based quadruple therapy and device evaluation. For HFmrEF, it mandates urgent OMT initiation and serial echocardiographic follow-up. For HFpEF, it directs comorbidity-centred management and emerging pharmacotherapy including SGLT2 inhibitors. Critically, the framework should not be applied as a static label: phenotypic reassessment every 3-6 months under optimally treated conditions is recommended to guide therapy titration and device decision-making
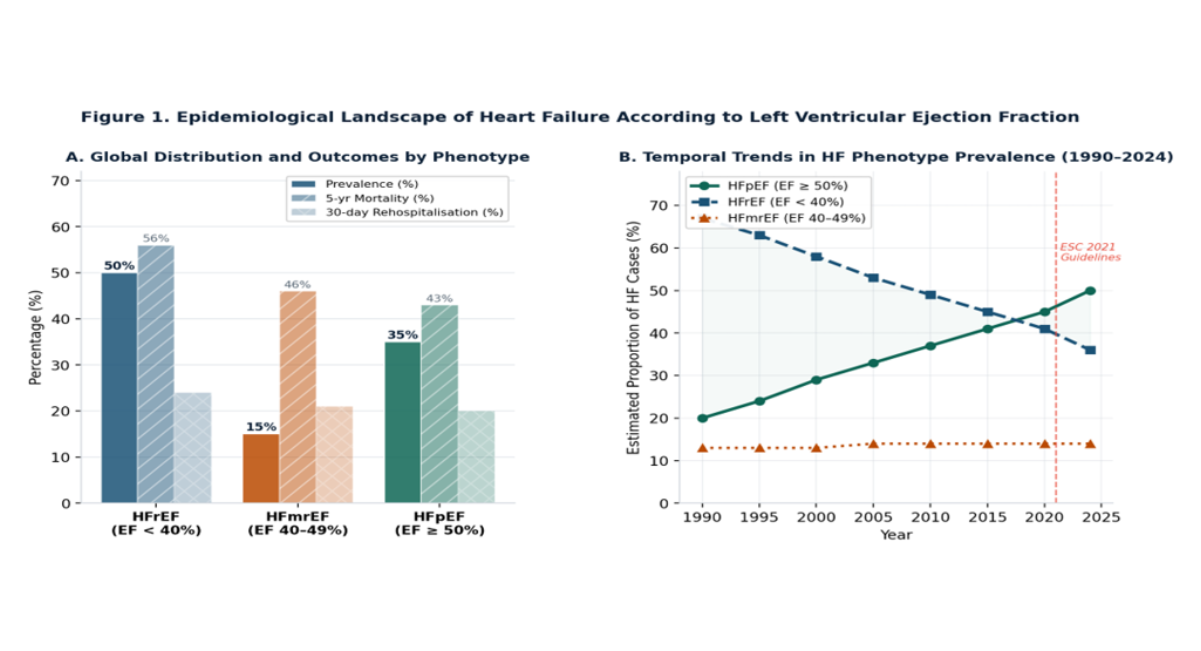
Figure 1: Epidemiological landscape of heart failure according to LVEF phenotype.
Panel A: prevalence, 5-year mortality, and 30-day rehospitalisation by phenotypic category.
Panel B: temporal trends in phenotype distribution from 1990 to 2024.
HFpEF = HF with preserved ejection fraction; HFrEF = HF with reduced ejection fraction; HFmrEF = HF with mildly reduced ejection fraction.
HFrEF: The Archetype, Its Natural History and the Biology of Deterioration
HFrEF remains the phenotype about which we know the most, treat most aggressively, and understand most deeply at a mechanistic level. Its natural history, absent treatment, follows relentless neurohumoral activation, progressive cardiomyocyte loss, adverse ventricular remodelling, and ultimately pump failure or sudden death. Landmark trials of neurohumoral blockade from the late 1980s through to DAPA-HF in 2019 have progressively modified this trajectory [9,12].
In contemporary optimally treated cohorts, 5-year mortality approximates 40-45% — measurably better than pre-trial populations. Mode of death has shifted: a 44% decline in sudden cardiac death risk between 1995 and 2014 reflects successful adoption of ICDs and neurohumoral blockade. What remains is an increasing proportion of deaths from pump failure and non-cardiovascular causes, with implications for future trial design [5].
The pathophysiology of HFrEF centres on RAAS activation, elevated noradrenaline, and heightened sympathetic tone collectively driving ventricular dilatation, mitral regurgitation, arrhythmia, and a vicious cycle of volume and pressure overload. Interstitial fibrosis stiffens the ventricular wall and impairs both systolic shortening and diastolic relaxation [9]. Of particular clinical relevance is the concept of reverse cardiac remodelling under OMT: between 35 and 40% of HFrEF patients will achieve HFrecEF if given an adequate duration of maximally tolerated OMT [8].
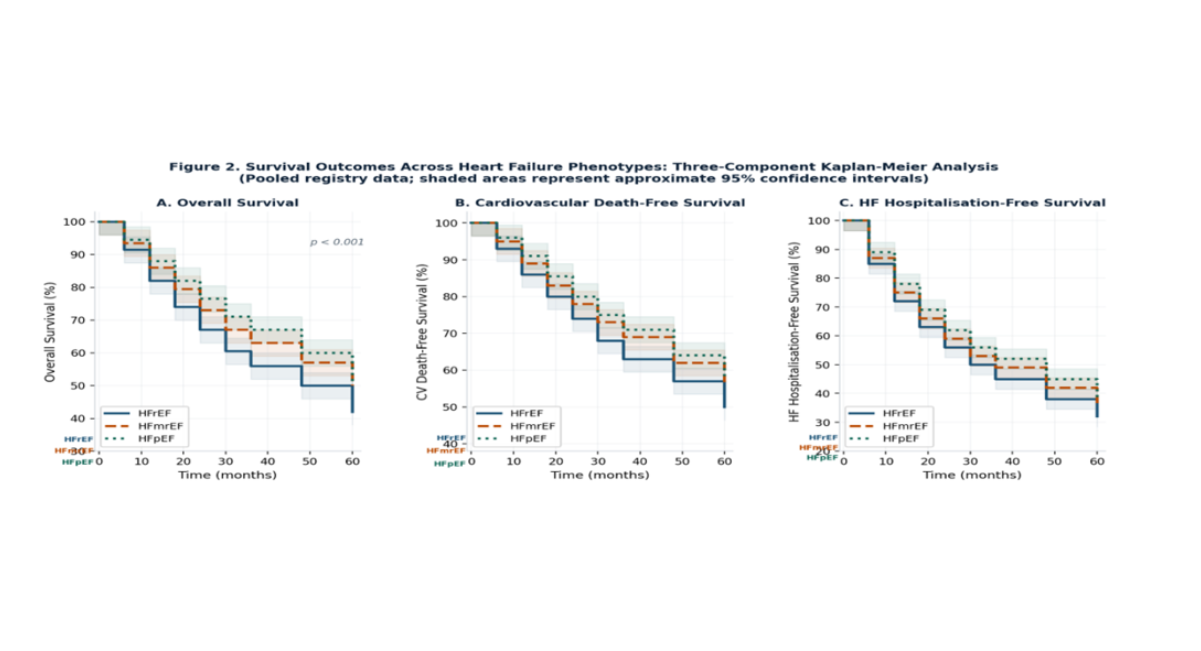
Figure 2: Three-component Kaplan-Meier analysis of survival outcomes across HF phenotypes.
Panel A: overall survival.
Panel B: cardiovascular death-free survival.
Panel C: HF hospitalisation-free survival. Shaded areas represent approximate 95% confidence intervals. p < 0.001 by log-rank test for all three endpoints (pooled registry data, representative estimates).
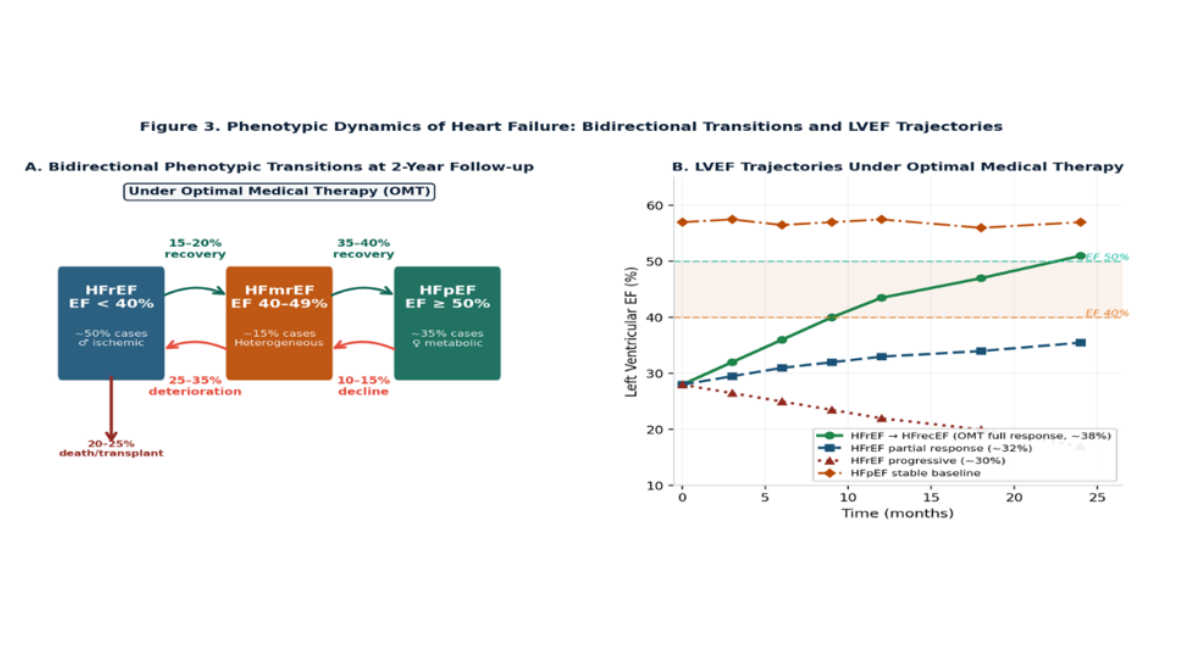
Figure 3: Phenotypic dynamics in heart failure
HFmrEF: The Dynamic Intermediate and Its Clinical Implications
HFmrEF is not merely a transitional state awaiting reclassification it is a phenotypically active state with a distinct biological character and a clinical trajectory acutely sensitive to early therapeutic choices. The data compellingly support a shift away from watchful waiting toward immediate, aggressive OMT initiation [6].
The demographic profile of HFmrEF patients sits between the two extremes: older than typical HFrEF patients but younger than HFpEF patients; ischaemic aetiology is common; atrial fibrillation and hypertension are frequent. A mixed picture of systolic impairment and diastolic dysfunction prevails which is precisely why the trajectory can tip in either direction [6,7].
Bhambhani and colleagues found that fewer than 30% of patients remained in the HFmrEF category at two-year follow-up [6]. The 35-40% who transition toward HFpEF largely do so because of LVEF recovery; the 25-35% who deteriorate toward HFrEF do so in the context of progressive neurohormonal activation, worsening fibrosis, and most importantly inadequate pharmacological optimisation. This last point is modifiable.
Patients who transition from HFmrEF to HFrEF may carry a worse prognosis than de novo HFrEF, reflecting more aggressive myocardial disease. Conversely, treatment-driven transition from HFmrEF to HFpEF confers a substantially more favourable prognosis than de novo HFpEF through hypertensive remodelling [3,8].
|
Step |
Action |
Timeframe |
|
Confirm diagnosis |
Echocardiography under stable, euvolaemic conditions; repeat if performed during decompensation |
At presentation |
|
Initiate OMT |
Start SGLT2i + beta-blocker; add RAAS inhibitor (ACE-i/ARB/ARNI) if LVEF < 45% or ischaemic aetiology |
Within 1-2 weeks |
|
Titrate to target doses |
Up-titrate all agents over 4-8 weeks; monitor BP, renal function, and potassium |
4-8 weeks |
|
Reassess LVEF |
Repeat echocardiography under optimised therapy to determine trajectory direction |
3-6 months |
|
Reclassify & adapt therapy |
If LVEF < 40%: escalate to full HFrEF OMT + device evaluation; If LVEF ≥ 50%: transition to HFpEF pathway |
Per reassessment |
|
Ongoing surveillance |
6-monthly echocardiography + biomarker monitoring; re-evaluate OMT at each visit |
Long-term |
Box 1: Proposed Clinical Algorithm for HFmrEF Management
Abbreviations: OMT = Optimal Medical Therapy; SGLT2i = SGLT2 Inhibitor; RAAS = Renin-Angiotensin-Aldosterone System; ACE-i = ACE Inhibitor; ARB = Angiotensin Receptor Blocker; ARNI = Angiotensin Receptor-Neprilysin Inhibitor; BP = Blood Pressure
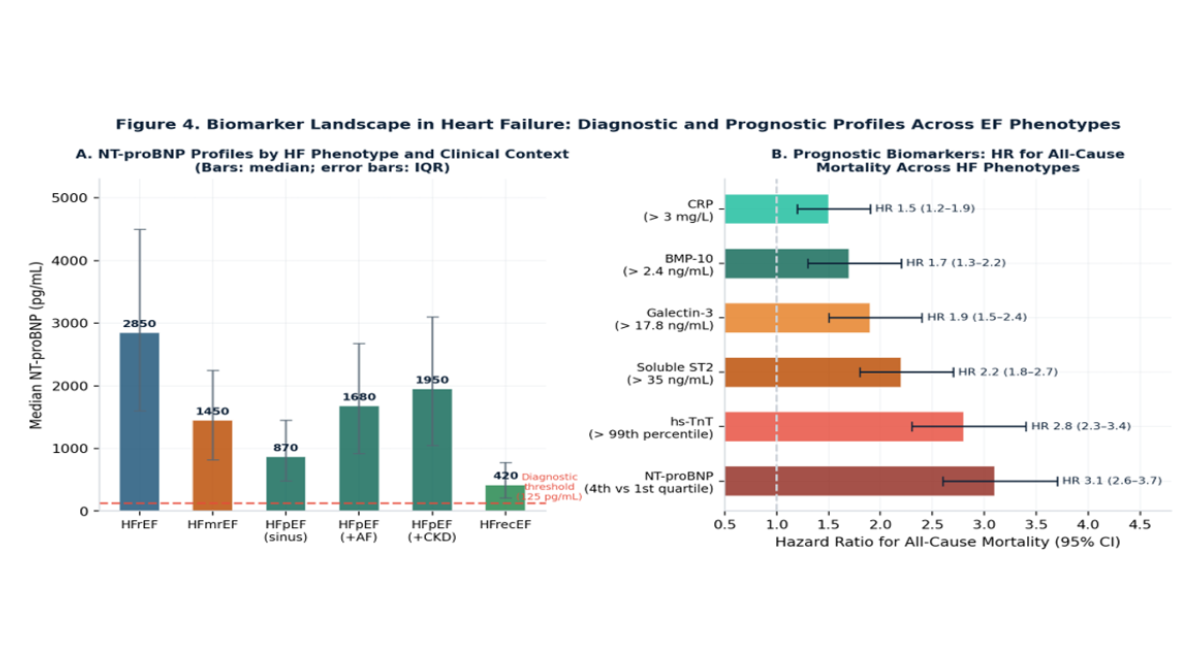
Figure 4: Biomarker landscape in heart failure.
Panel A: NT-proBNP profiles by HF phenotype and clinical context; bars represent medians with IQR error bars; the dashed line indicates the 125 pg/mL ESC diagnostic threshold.
Panel B: hazard ratios (95% CI) for all-cause mortality associated with established prognostic biomarkers across HF phenotypes.
Abbreviations: AF = Atrial Fibrillation; CKD = Chronic Kidney Disease; HFrecEF = HF with recovered EF.
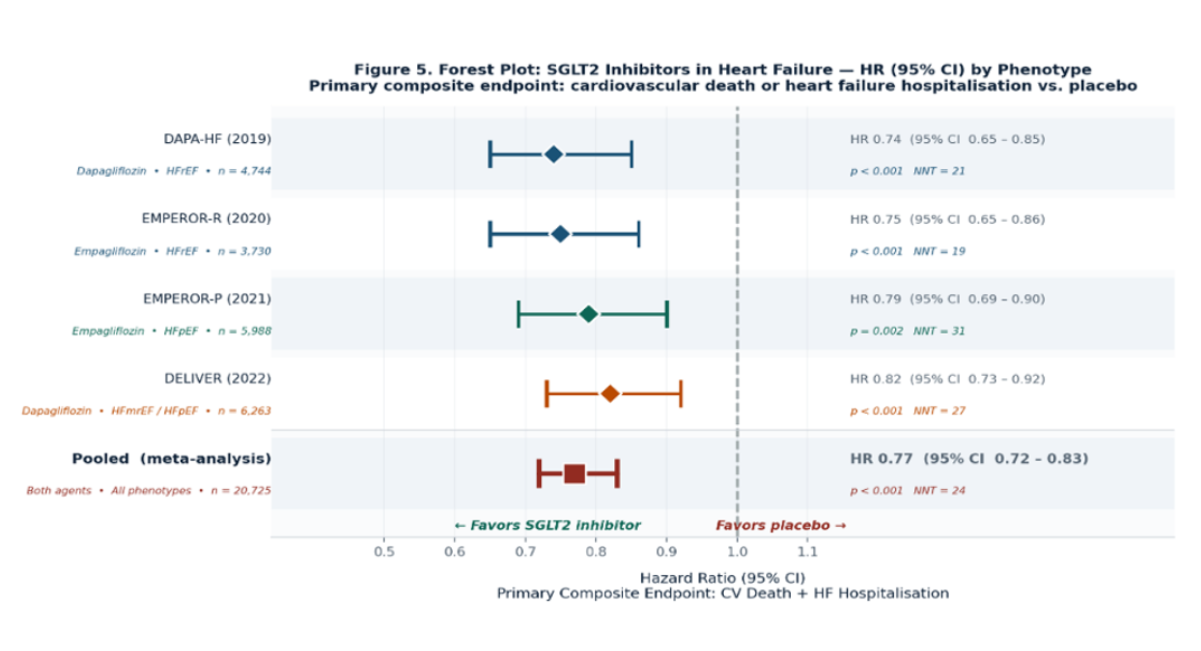
Figure 5: Forest plot of SGLT2 inhibitor randomised controlled trials across heart failure phenotypes. Primary composite endpoint: cardiovascular death or HF hospitalisation. HR values are taken from primary publications. NNT = number needed to treat; CI = confidence interval. Pooled HR from Zannad [12-16].
Therapeutic Trajectories: Optimisation, Remodelling and the Sglt2 Revolution
OMT in HFrEF: A Quadruplet That Saves Lives
The pharmacological management of HFrEF rests on four drug classes RAAS inhibitors (ACE inhibitors, ARBs, or ARNI), beta-blockers, mineralocorticoid receptor antagonists (MRAs), and SGLT2 inhibitors each with robust RCT-derived mortality evidence targeting a distinct arm of neurohormonal-metabolic dysregulation. When all four are combined at target doses, modelling estimates suggest an absolute mortality reduction of 40-60% relative to diuresis-only therapy [4,17].
Therapeutic inertia remains deeply persistent: fewer than 25% of HFrEF patients discharged from hospital receive target doses of all guideline-recommended classes [17]. The ARNI sacubitril/valsartan reduced cardiovascular death and HF hospitalisation by 20% relative to enalapril (HR 0.80, 95% CI 0.73-0.87) in PARADIGM-HF [4], with echocardiographic sub-studies confirming greater LVEF recovery and LV volume regression than enalapril alone [8]. Full four-class OMT is associated with a HFrecEF probability of approximately 40%, compared with under 10% for monotherapy.
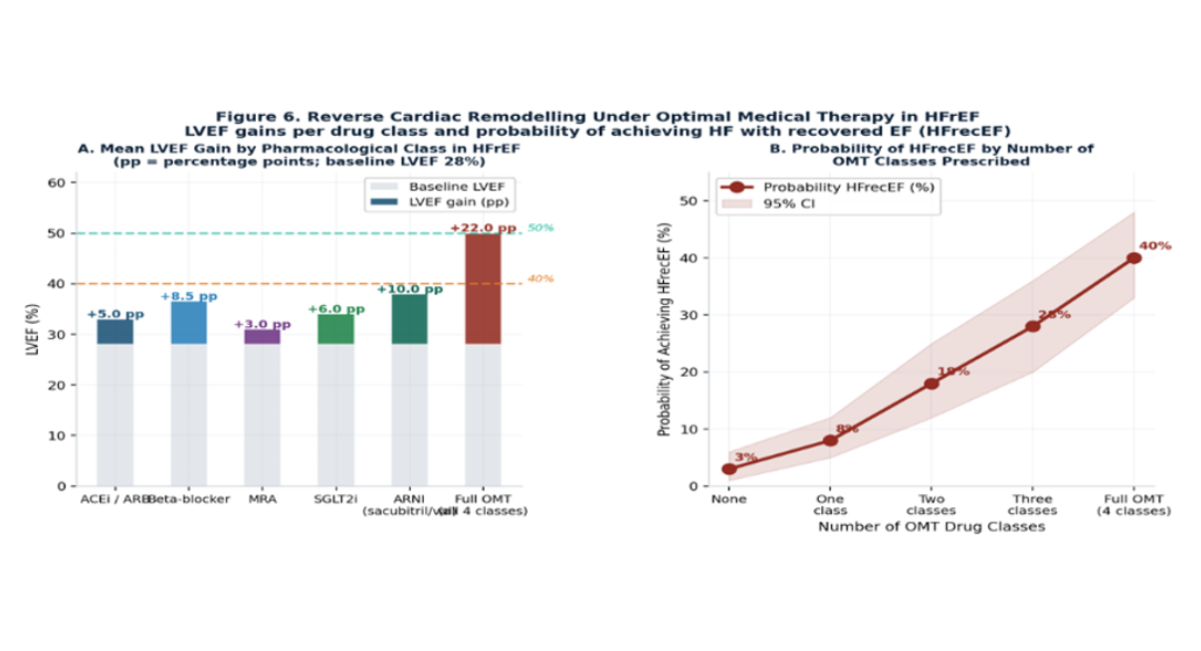
Figure 6: Reverse cardiac remodelling under optimal medical therapy in HFrEF.
Panel A: mean LVEF gain (percentage points above a 28% baseline) by pharmacological class.
Panel B: probability of achieving HF with recovered ejection fraction (HFrecEF) by number of OMT drug classes prescribed.
Abbreviations: PP = Percentage Points; ARNI = Angiotensin Receptor–Neprilysin Inhibitor; MRA = Mineralocorticoid Receptor Antagonist; SGLT2i = SGLT2 Inhibitor.
HFpEF: Pathophysiological Complexity and the Comorbidity Matrix
Pathophysiology
HFpEF is the most biologically complex entity in contemporary cardiology. Its pathophysiology begins not in the ventricle but in the systemic microvasculature. Excess comorbidities hypertension, obesity, diabetes, sleep apnoea drive systemic pro-inflammatory activation that damages the coronary microvascular endothelium, reduces nitric oxide bioavailability, and stimulates interstitial collagen deposition [9]. The result is a stiff, thick-walled, fibrotic ventricle, impaired in its ability to relax at normal filling pressures. Symptoms arise from an inability to fill rather than an inability to eject a physiology particularly exposed during exercise [9,11].
Phenotypic Heterogeneity
The phenotypic heterogeneity within HFpEF is considerable. Tromp and colleagues, applying latent class analysis in a multi-ethnic Asian cohort, identified four biologically coherent sub-phenotypes: (1) a metabolic-dominant cluster (obesity, diabetes, ~40% of cases); (2) a cardiorenal cluster (~20%); (3) a right ventricular failure and atrial fibrillation cluster (~20%); and (4) a mixed infiltrative/elderly cluster (~20%) [10]. These clusters carry different prognoses and may ultimately require different therapeutic approaches.
Prognosis
The prognostic profile of HFpEF has been substantially reappraised. Earlier registry data showing better survival in HFpEF than HFrEF reflected differences in age, sex, and comorbidity distribution rather than genuine cardiac advantage. Contemporary analyses show broadly equivalent cardiovascular mortality across EF categories, with the additional burden of a high non-cardiovascular death rate in HFpEF particularly from malignancy and renal failure [3]. This non-cardiovascular mortality is mechanistically linked to the same inflammatory-metabolic milieu driving the cardiac pathology.
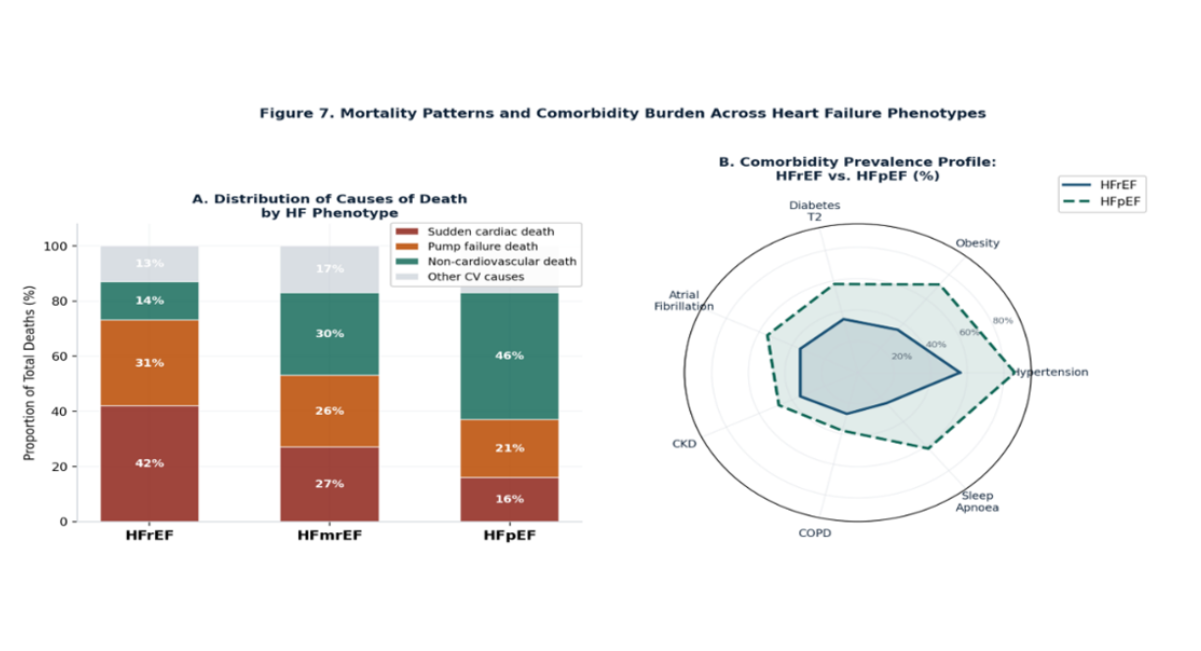
Figure 7: Mortality patterns and comorbidity burden across heart failure phenotypes
Biomarker-Guided Risk Stratification Across Phenotypes
Biomarker assessment in HF serves multiple distinct roles: diagnosis, prognostic stratification, therapeutic monitoring, and phenotypic sub-classification. NT-proBNP retains diagnostic utility across all three EF categories (threshold: 125 pg/mL in non-acute settings), with median values that differ substantially between phenotypes: exceeding 2,500 pg/mL in HFrEF versus 800-1,200 pg/mL in HFpEF with sinus rhythm and preserved renal function [1,4,11].
Key confounders of NT-proBNP interpretation include: atrial fibrillation (raises values substantially), obesity (suppresses via adipokine-mediated clearance), and severe renal impairment (inflates values through fragment accumulation). These confounders are clinically important given their co-prevalence exceeds 50% in HFpEF patients. Serial NT-proBNP changes under therapy track clinical improvement in a way that symptoms alone cannot capture. Beyond natriuretic peptides, the prognostic landscape has been enriched by high-sensitivity troponin T, soluble ST2, and galectin-3, each capturing a distinct dimension of myocardial pathobiology cardiomyocyte injury, inflammation and fibrosis signalling, and extracellular matrix turnover, respectively [4,11]. Multi-biomarker profiling, rather than reliance on any single marker, is most likely to capture the multi-dimensional disease biology a principle informing the ongoing development of integrated risk scores combining echocardiographic, laboratory, and patient-reported parameters.
SGLT2 Inhibitors: Cross-Phenotype Efficacy and Mechanistic Pluralism
SGLT2 inhibitors represent the most consequential pharmacological advance in HF since the introduction of beta-blockade. What makes this advance particularly noteworthy is not simply the magnitude of benefit a 23-26% relative risk reduction in hard HF outcomes — but that this benefit extends across phenotypes in a way no prior drug class has managed.
DAPA-HF (2019) enrolled 4,744 HFrEF patients and demonstrated that dapagliflozin 10 mg daily reduced the composite of CV death, HF hospitalisation, or urgent HF visit by 26% (HR 0.74, 95% CI 0.65-0.85), with identical benefit across diabetes status [12]. EMPEROR-R (2020) confirmed these findings with empagliflozin, adding a signal of slowed eGFR decline [13]. EMPEROR-P (2021) and DELIVER (2022) extended efficacy to HFpEF and HFmrEF [14,15].
Multiple non-exclusive mechanisms explain cross-phenotype efficacy: osmotic diuresis and natriuresis reducing preload; epicardial adipose tissue reduction attenuating myocardial inflammation; inhibition of the sodium-hydrogen exchanger in cardiomyocytes; renal tubuloglomerular feedback reducing intraglomerular hypertension; and provision of beta-hydroxybutyrate as an energetically favourable alternative substrate [12,16]. The multiplicity of partially independent mechanisms likely explains the universality of benefit.
|
Trial (Year) |
Drug |
HF Phenotype |
N enrolled |
Primary HR (95% CI) |
p-value |
NNT |
|
DAPA-HF (2019) |
Dapagliflozin 10 mg |
HFrEF (EF < 40%) |
4,744 |
0.74 (0.65-0.85) |
< 0.001 |
21 |
|
EMPEROR-R (2020) |
Empagliflozin 10 mg |
HFrEF (EF < 40%) |
3,730 |
0.75 (0.65-0.86) |
< 0.001 |
19 |
|
EMPEROR-P (2021) |
Empagliflozin 10 mg |
HFpEF (EF ≥ 40%) |
5,988 |
0.79 (0.69-0.90) |
0.002 |
31 |
|
DELIVER (2022) |
Dapagliflozin 10 mg |
HFmrEF/HFpEF (EF ≥ 40%) |
6,263 |
0.82 (0.73-0.92) |
< 0.001 |
27 |
|
Zannad meta-analysis (2020) |
Both agents |
All phenotypes |
20,725 |
0.77 (0.72-0.83) |
< 0.001 |
24 |
Table 2: Summary of Major SGLT2 Inhibitor Trials in Heart Failure across EF Phenotypes
Abbreviations: HR = hazard ratio; CI = confidence interval; NNT = number needed to treat at median follow-up.
Emerging Pharmacological Therapies beyond SGLT2 Inhibitors
The therapeutic armamentarium for HF continues to expand. Two emerging drug classes warrant emphasis for their growing evidence base:
Finerenone, a non-steroidal mineralocorticoid receptor antagonist (MRA) with a more selective receptor binding profile than spironolactone, demonstrated a 16% relative reduction in HF events in the FINEARTS-HF trial (2024) across the HFmrEF and HFpEF spectrum. Its lower hyperkalaemia risk and superior renal tolerability compared to steroidal MRAs position it as a candidate for broader use in patients with HFpEF and chronic kidney disease a population in whom spironolactone is frequently withheld due to renal safety concerns [18].
Vericiguat, a soluble guanylate cyclase stimulator targeting the nitric oxide-cGMP pathway, has shown benefit in high-risk HFrEF patients with a recent worsening event (VICTORIA trial; HR 0.90, 95% CI 0.82-0.98) [18,19]. By restoring impaired NO-cGMP signalling in the failing myocardium a mechanism distinct from neurohumoral blockade vericiguat may offer additive benefit in the subset of the progressive HFrEF trajectory subgroup that fails to respond adequately to conventional OMT. These agents expand the therapeutic landscape and should be considered in appropriately selected patients as evidence continues to mature.
Hospitalisation, Readmission and the Challenge of the "Revolving Door"
Heart failure hospitalisation is a prognostic inflection point, associated with accelerated myocardial damage, worsening renal function, progressive deconditioning, and a measurable increment in risk of the next admission and death. The 30-day readmission rate of 20-25% across HF phenotypes has changed little despite extensive quality improvement initiatives [4,17].
Predictors of early readmission include: residual NT-proBNP above 1,000 pg/mL at discharge, failure to initiate or up-titrate OMT before discharge, inadequate diuresis, impaired renal function, and absence of structured follow-up within seven days. Machine learning-based risk stratification tools show promise: AUC values of 0.78-0.85 for 30-day readmission prediction have been reported from models integrating clinical, laboratory, and echocardiographic parameters [18].
Mortality Patterns and the Implications for Trial Design
The heterogeneity of death modes across HF phenotypes has direct implications for future trial design. The dominance of SCD in HFrEF (42% of total deaths) explains why ICD therapy produces its largest absolute benefit in this population. In HFpEF, the high proportion of non-cardiovascular deaths (46%, predominantly malignancy and renal failure) raises a philosophical question: a composite of CV death and HF hospitalisation may systematically underestimate total disease burden in HFpEF. Future HFpEF trials should consider all-cause mortality or broader composite endpoints to avoid inadvertent underestimation of treatment effect.
DISCUSSION
The central argument of this review can be stated simply: heart failure is a moving target. The EF category assigned at a single clinical encounter is a snapshot of a biological process continuously evolving in response to treatment, comorbidity progression, and intrinsic myocardial disease. The clinical and prognostic consequences of movement between categories are substantial enough to demand active monitoring rather than assumed stability.
HFmrEF should be treated as a therapeutic emergency in disguise. The window during which aggressive OMT may arrest or reverse phenotypic deterioration is finite, and inaction has measurable downstream consequences: HFrEF progression, reduced LVEF recovery probability, and higher long-term mortality [6,17]. The clinical culture of cautious, incremental pharmacological optimisation appropriate in many conditions is counterproductive in HFmrEF, where the rate of phenotypic transition is fastest and the impact of early, full OMT is greatest.
Patients classified as HFpEF should not be assumed to have arrived at that classification from the same biological starting point. A patient whose HFpEF follows treatment-driven LVEF recovery from prior HFrEF carries a fundamentally different biological substrate from one whose HFpEF represents the natural culmination of hypertensive remodelling [3,8]. This distinction matters for prognosis and may ultimately matter for therapy.
The emergence of SGLT2 inhibitors as a cross-phenotype therapy represents a genuine paradigm shift. No prior drug class had demonstrated mortality-relevant efficacy across the full EF spectrum. What SGLT2 inhibitors have done is circumvent EF-based therapeutic stratification, simplifying and accelerating prescribing in a disease where therapeutic inertia historically costs lives [17].
As this review also demonstrates, the therapeutic landscape is expanding beyond SGLT2 inhibitors. Finerenone and vericiguat represent distinct mechanistic approaches with emerging evidence in specific HF subpopulations, and gene therapies targeting calcium handling proteins are progressing through early-phase evaluation [18,19,20]. These developments will further refine trajectory-informed prescribing.
Several limitations of this review merit acknowledgement. As a narrative synthesis, it does not provide the quantitative precision of a formal meta-analysis. The represented epidemiological and trial data derive predominantly from high-income European and North American populations, with limited representation from sub-Saharan African settings where aetiological patterns differ substantially. Future registry data from resource-limited settings will be essential to determine the generalisability of these findings.
Looking ahead, precision phenotyping using multi-omic data proteomic signatures, transcriptomic profiles, cardiac MRI-based tissue characterisation is progressively moving from research settings toward clinical feasibility. Machine learning models can already predict two-year phenotypic transitions from HFmrEF with an AUC of 0.82-0.86 [18], outperforming single-variable predictors including LVEF itself.
CONCLUSION
This narrative review of HF evolutionary trajectories across the EF spectrum yields four key take-home messages:
|
# |
Key Take-Home Message |
|
1 |
LVEF must be measured serially, under stable and optimally treated conditions. Phenotypic stability is the exception, not the rule — particularly in HFmrEF (>70% two-year transition probability) and HFrEF (~40% HFrecEF probability under full OMT). |
|
2 |
HFmrEF demands the same urgency as HFrEF. The window for pharmacological intervention is finite; the cost of inaction is measurable in HFrEF progression, lost LVEF recovery probability, and excess mortality. Deferred prescribing in this category is not supported by evidence. |
|
3 |
HFpEF is not a single disease. Its four biologically coherent sub-phenotypes carry different prognoses and may ultimately require different therapeutic strategies. The arrival of SGLT2 inhibitors as proven therapy is a landmark, but likely the beginning of phenotype-specific differentiation, not the endpoint. |
|
4 |
SGLT2 inhibitors are the only drug class with demonstrated disease-modifying efficacy across all three HF phenotypes. Their multi-pathway mechanism supports early, universal prescription in all patients with symptomatic HF who lack absolute contraindications, independent of EF category. |
The future of HF management lies in dynamic, trajectory-informed precision phenotyping — integrating serial echocardiographic data, multi-biomarker profiles, and machine learning-based risk stratification to anticipate phenotypic transitions and adapt therapy accordingly. This vision is moving from aspiration to feasibility.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Not applicable. This article is a narrative review of previously published data; no primary data collection was involved.
CONFLIT OF INTRESTS
The author declares no financial or non-financial competing interests relevant to this manuscript.
FUNDING
This work received no external funding.
AUTHOR CONTRIBUTIONS
Manyebwa Kalemera Jean de Dieu: conceptualisation, systematic literature search, data synthesis and interpretation, writing of original draft, critical revision, and approval of the final manuscript.
REFERENCES
- McDonagh T, Metra M. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. Russian Journal of Cardiology. 2023;28(1):5168. [Crossref] [Google Scholar] [PubMed]
- Savarese G, Lund LH. Global public health burden of heart failure. Cardiac failure review. 2017;3(1):7. [Crossref] [Google Scholar] [PubMed]
- Lam CS, Solomon SD. Classification of heart failure according to ejection fraction: JACC review topic of the week. Journal of the American College of Cardiology. 2021;77(25):3217-25. [Crossref] [Google Scholar] [PubMed]
- Borlaug BA. Evaluation and management of heart failure with preserved ejection fraction. Nature Reviews Cardiology. 2020;17(9):559-73. [Crossref] [Google Scholar] [PubMed]
- Shen L, Jhund PS, Petrie MC, Claggett BL, Barlera S, Cleland JG, et al. Declining risk of sudden death in heart failure. New England Journal of Medicine. 2017;377(1):41-51. [Crossref] [Google Scholar] [PubMed]
- Bhambhani V, Kizer JR, Lima JAC, Van Der Harst P, Bahrami H, Nayor M, et al. Predictors and outcomes of heart failure with mid-range ejection fraction. Eur J Heart Fail. 2018;20(4):651-659. [Crossref] [Google Scholar] [PubMed]
- Lauritsen J, Gustafsson F, Abdulla J. Characteristics and long-term prognosis of patients with heart failure and mid-range ejection fraction compared with reduced and preserved ejection fraction: a systematic review and meta-analysis. ESC heart failure. 2018;5(4):685-94. [Crossref] [Google Scholar] [PubMed]
- Cleland JG, Bunting KV, Flather MD, Altman DG, Holmes J, Coats AJ, et al. Beta-blockers for heart failure with reduced, mid-range, and preserved ejection fraction: an individual patient-level analysis of double-blind randomized trials. European heart journal. 2018;39(1):26-35. [Crossref] [Google Scholar] [PubMed]
- Braunwald E. The war against heart failure: the Lancet lecture. The Lancet. 2015;385(9970):812-24. [Crossref] [Google Scholar] [PubMed]
- Tromp J, MacDonald MR, Tay WT, Teng TH, Hung CL, Narasimhan C, et al. Heart failure with preserved ejection fraction in the young. Circulation. 2018;138(24):2763-73. [Crossref] [Google Scholar] [PubMed]
- Pieske B, Tschöpe C, De Boer RA, Fraser AG, Anker SD, Donal E, et al. How to diagnose heart failure with preserved ejection fraction: the HFA–PEFF diagnostic algorithm: a consensus recommendation from the Heart Failure Association (HFA) of the European Society of Cardiology (ESC). European heart journal. 2019;40(40):3297-317. [Crossref] [Google Scholar] [PubMed]
- McMurray JJ, Solomon SD, Inzucchi SE, Køber L, Kosiborod MN, Martinez FA, et al. Dapagliflozin in patients with heart failure and reduced ejection fraction. New England Journal of Medicine. 2019;381(21):1995-2008. [Crossref] [Google Scholar] [PubMed]
- Packer M, Anker SD, Butler J, Filippatos G, Pocock SJ, Carson P, et al. (EMPEROR-R) Cardiovascular and renal outcomes with empagliflozin in heart failure. N Engl J Med. 2020;383(15):1413-1424. [Crossref] [Google Scholar] [PubMed]
- Anker SD, Butler J, Filippatos G, Ferreira JP, Bocchi E, Böhm M, et al. Empagliflozin in heart failure with a preserved ejection fraction. New England Journal of Medicine. 2021;385(16):1451-61. [Crossref] [Google Scholar] [PubMed]
- Solomon SD, McMurray JJ, Claggett B, de Boer RA, DeMets D, Hernandez AF, et al. Dapagliflozin in heart failure with mildly reduced or preserved ejection fraction. New England Journal of Medicine. 2022;387(12):1089-98. [Crossref] [Google Scholar] [PubMed]
- Zannad F, Ferreira JP, Pocock SJ, et al. SGLT2 inhibitors in patients with heart failure with reduced ejection fraction: a meta-analysis of the EMPEROR-R and DAPA-HF trials. Lancet. 2020;396(10254):819-829. [Crossref] [Google Scholar] [PubMed]
- Greene SJ, Fonarow GC, DeVore AD, et al. Titration of medical therapy for heart failure with reduced ejection fraction. J Am Coll Cardiol. 2019;73(19):2365-2383. [Crossref] [Google Scholar] [PubMed]
- Borlaug BA, Sharma K, Shah SJ, Ho JE. Heart failure with preserved ejection fraction: JACC scientific statement. Journal of the American College of Cardiology. 2023;81(18):1810-34. [Crossref] [Google Scholar] [PubMed]
- Seferovic PM, Ponikowski P, Anker SD, Bauersachs J, Chioncel O, Cleland JG, et al. Clinical practice update on heart failure 2019: pharmacotherapy, procedures, devices and patient management. An expert consensus meeting report of the Heart Failure Association of the European Society of Cardiology. European journal of heart failure. 2019;21(10):1169-86. [Crossref] [Google Scholar] [PubMed]
- Lam CS, Voors AA, De Boer RA, Solomon SD, Van Veldhuisen DJ. Heart failure with preserved ejection fraction: from mechanisms to therapies. European heart journal. 2018;39(30):2780-92. [Crossref] [Google Scholar] [PubMed]
Article Processing Timeline
| 2-5 Days | Initial Quality & Plagiarism Check |
| 15 Days |
Peer Review Feedback |
| 85% | Acceptance Rate (after peer review) |
| 30-45 Days | Total article processing time |
Journal Flyer